Quaternary (chemistry)
|
Central atoms marked in red in various groups of substances . Comparison of quaternary with primary , secondary and tertiary central atoms. |
||||
| primary | secondary | tertiary | quaternary | |
|
Carbon atom of an organic compound |

|

|

|

|
| alcohol |

|
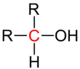
|

|
does not exist |
| Amine |

|

|

|
 (see QAV ) |
| Carboxamide |

|

|

|
does not exist |
| Phosphine |

|

|
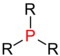
|
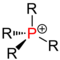 (see QPV ) |
In organic chemistry , the word "quaternary" is a name for four organic radicals (organyl radical, such as alkyl radical, alkenyl radical, aryl radical, alkylaryl radical, etc. ) bound to a central atom ( carbon , nitrogen , phosphorus ) . ).
Examples of quaternary central atoms
- a carbon atom in an alkane to which four substituents but no hydrogen atom are bonded,
- a nitrogen atom in a quaternary ammonium salt to which four carbon atoms are bonded or
- a phosphorus atom in a quaternary phosphonium salt to which four carbon atoms are bonded.
Examples of such compounds are the alkane neopentane ( 2,2-dimethylpropane ) with the quaternary carbon atom in the middle, the quaternary ammonium salt tetrabutylammonium bromide and the quaternary phosphonium salt tetramethylphosphonium bromide .
See also
Individual evidence
- ↑ According to the IUPAC nomenclature, secondary or tertiary amides are substituted with two or three acyl groups on the nitrogen. However, since primary amides with one acyl group and two hydrocarbon substituents on the nitrogen are called "tertiary amides" in common usage, classification into primary, secondary and tertiary amides is not recommended. Entry to amides. In: Römpp Online . Georg Thieme Verlag, accessed on May 15, 2019.
- ^ Brockhaus ABC Chemie , VEB FA Brockhaus Verlag Leipzig 1965, p. 1274.
- ↑ Otto-Albrecht Neumüller (Ed.): Römpps Chemie-Lexikon. Volume 6: T-Z. 8th revised and expanded edition. Franckh'sche Verlagshandlung, Stuttgart 1988, ISBN 3-440-04516-1 , p. 4170.