Titanium (III) sulfide
| Crystal structure | |||||||
|---|---|---|---|---|---|---|---|
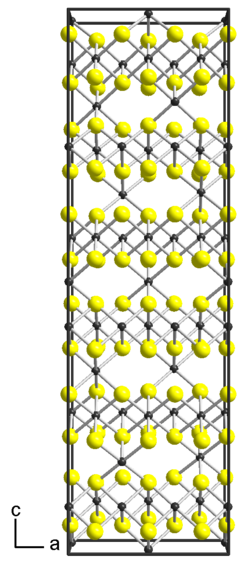
|
|||||||
| __ Ti 3+ __ S 2− | |||||||
| General | |||||||
| Surname | Titanium (III) sulfide | ||||||
| other names |
Dititanium trisulfide |
||||||
| Ratio formula | Ti 2 S 3 | ||||||
| Brief description |
black powder |
||||||
| External identifiers / databases | |||||||
|
|||||||
| properties | |||||||
| Molar mass | 191.93 g mol −1 | ||||||
| Physical state |
firmly |
||||||
| density |
3.684 g cm −3 |
||||||
| safety instructions | |||||||
|
|||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||
Titanium (III) sulfide or dititanium trisulfide , formerly also called titanium sesquisulfide according to its ratio formula , is a chemical compound from the group of sulfides with the formula Ti 2 S 3 .
presentation
Titanium (III) sulfide is produced from titanium (IV) sulfide TiS 2 by either heating it to 1000 ° C in a vacuum or reducing it with hydrogen at high temperatures. Dititanium trisulfide can also be produced from the elements under pressure and at 800 ° C.
properties
Titanium (III) sulfide is a black powder that can also be finely crystalline or shiny. The structure in the crystal is related to the nickel arsenide structure (hexagonal closest packing of anions), with a coordination number of titanium of 6.
Titanium (III) sulfide is stable in air and water at normal temperatures and therefore, unlike titanium disulfide, does not smell of hydrogen sulfide.
In hot sulfuric acid, Ti 2 S 3 initially forms a blue-gray turbidity, then finally a colorless solution, while in cold, concentrated sulfuric or nitric acid, green-colored solutions are formed. Hydrogen sulfide is formed with hot hydrochloric acid.
Web links
- Ping Yang: Photoluminescence characteristics of Ti2S3 nanoparticles embedded in sol – gel derived silica xerogel (PDF; 162 kB)
Individual evidence
- ^ A b c d A. F. Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1525.
- ^ Jean D'Ans, Ellen Lax: Pocket book for chemists and physicists . 2007, ISBN 978-3-540-60035-0 ( page 770 in the Google book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ a b c d Gmelin's Handbook of Inorganic Chemistry, Titan