Tolprocarb
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
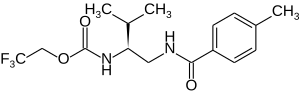
|
|||||||||||||
| General | |||||||||||||
| Surname | Tolprocarb | ||||||||||||
| other names |
2,2,2-trifluoroethyl- N - [( 1S ) -2-methyl-1 - ([(4-methylbenzoyl) amino] methyl]) propyl carbamate |
||||||||||||
| Molecular formula | C 16 H 21 F 3 N 2 O 3 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 346.34 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Tolprocarb a chemical compound from the group of carbamates , which is effective as a fungicide against Magnaporthe grisea . The compound was developed by Mitsui Chemicals .
Tolprocarb works by inhibiting polyketide synthase in melanin biosynthesis.
Admission
Tolprocarb is not approved as a crop protection agent in the European Union or Switzerland .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ a b Takahiro Hamada, Mineko Asanagi, Tomomi Satozawa, Natsuko Araki, Shinichi Banba, Norikazu Higashimura, Tomohisa Akase, Kangetsu Hirase: Action mechanism of the novel rice blast fungicide tolprocarb distinct from that of conventional melanin biosynthesis inhibitors . In: Journal of Pesticide Science . tape 39 , no. 3 , June 2014, p. 152 , doi : 10.1584 / jpestics.D14-033 (English, PDF ).
- ^ Directorate-General for Health and Food Safety of the European Commission: EU pesticide database ; Entry in the national directory of plant protection products in Switzerland ; Retrieved June 25, 2016.
