Ammonium magnesium phosphate
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
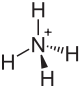 
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Ammonium magnesium phosphate | ||||||||||||||||||
| Molecular formula | NH 4 MgPO 4 | ||||||||||||||||||
| Brief description |
white crystals |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 137.32 g mol −1 (anhydrous) | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| solubility |
almost insoluble in water (0.2 g / L) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Ammonium magnesium phosphate is a salt made from ammonium and magnesium cations and phosphate anions with the formula (NH 4 MgPO 4 · x H 2 O). It is also known as triple phosphate .
Occurrence
The compound occurs as the mineral struvite in guano , for example , and often comes from human or animal waste. The connection is found in the urine sediment . Furthermore, ammonium magnesium phosphate can form during biogas production in dry fermentation plants.
properties
Ammonium magnesium phosphate forms colorless, water-insoluble, but acid-soluble, highly refractive crystals that appear in the coffin lid - or very rarely in the form of bracken .
use
Ammonium magnesium phosphate is formed during the detection reaction of magnesium ions (Mg 2+ ) with diammonium hydrogen phosphate .
Individual evidence
- ↑ a b c d data sheet ammonium magnesium phosphate hydrate from AlfaAesar, accessed on December 7, 2019 ( PDF )(JavaScript required) .
- ↑ Phillip Barak and Alysa Stafford: STRUVITE: A RECOVERED AND RECYCLED PHOSPHORUS FERTILIZER ( memento June 20, 2010 in the Internet Archive ), accessed April 2, 2014
- ↑ Giovanni B. Fogazzi al .: et urinalysis: Core Curriculum 2008 . In: American Journal of Kidney Diseases . Vol. 51, Issue 6, 2008, pp. 1052-1067 , doi : 10.1053 / j.ajkd.2007.11.039 ( article ).
- ^ E. Schweda: Jander / Blasius: Inorganic Chemistry I - Introduction & Qualitative Analysis . 17th edition. Hirzel, 2012, ISBN 978-3-7776-2134-0 , pp. 301 .

