UDP-glucose-4-epimerase
| UDP-glucose-4-epimerase | ||
|---|---|---|

|
||
| UDP-glucose-4-epimerase dimer (human) according to PDB 1EK5 | ||
|
Existing structural data: s. UniProt |
||
| Properties of human protein | ||
| Mass / length primary structure | 348 amino acids | |
| Secondary to quaternary structure | Homodimer | |
| Cofactor | NAD + | |
| Identifier | ||
| Gene name | GAEL | |
| External IDs | ||
| Enzyme classification | ||
| EC, category | 5.1.3.2 , isomerase | |
| Response type | Rearrangement | |
| Substrate | UDP-galactose, UDP- N -acetylgalactosamine | |
| Products | UDP-glucose, UDP- N -acetylglucosamine | |
| Occurrence | ||
| Homology family | NAD-dependent epimerase | |
| Parent taxon | Creature | |
| Orthologue | ||
| human | House mouse | |
| Entrez | 2582 | 74246 |
| Ensemble | ENSG00000117308 | ENSMUSG00000028671 |
| UniProt | Q14376 | Q8R059 |
| Refseq (mRNA) | NM_000403 | NM_178389 |
| Refseq (protein) | NP_000394 | NP_848476 |
| Gene locus | Chr 1: 23.8 - 23.8 Mb | Chr 4: 135.96 - 135.97 Mb |
| PubMed search | 2582 |
74246
|
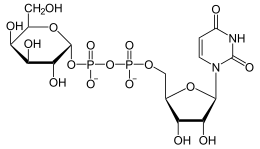
UDP-glucose 4-epimerase ( gene : GALE ), and UDP-galactose-4-epimerase , is the enzyme that the rearrangement of UDP-galactose to UDP-glucose and vice versa catalyzed . This reaction is a partial step in the utilization of galactose in many living things. Mutations in the GALE gene can lead to epimerase deficiency, which leads to type 3 galactosemia .
Catalyzed equilibrium
UDP-glucose is rearranged to UDP-galactose and vice versa. UDP- N -acetylglucosamine and -galactosamine are accepted as alternative substrates , but these are of no importance for the metabolism as a whole.
Web links
Wikibooks: Biochemistry and Pathobiochemistry: Galactose Metabolism - Learning and Teaching Materials
Individual evidence
- ↑ UniProt Q14376
- ↑ Schulz JM, Ross KL, Malmstrom K, Krieger M, Fridovich-Keil JL: Mediators of galactose sensitivity in UDP-galactose 4'-epimerase-impaired mammalian cells . In: J. Biol. Chem. . 280, No. 14, April 2005, pp. 13493-502. doi : 10.1074 / jbc.M414045200 . PMID 15701638 .