Vanadium (V) oxide trifluoride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
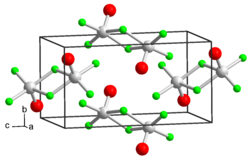
|
||||||||||||||||
| __ V 5+ __ F - __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Vanadium (V) oxide trifluoride | |||||||||||||||
| other names |
|
|||||||||||||||
| Ratio formula | VOF 3 | |||||||||||||||
| Brief description |
light yellow solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 123.94 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.459 g cm −3 |
|||||||||||||||
| Melting point |
300 ° C |
|||||||||||||||
| boiling point |
480 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Vanadium (V) oxide trifluoride is an inorganic chemical compound of vanadium from the group of oxide fluorides .
Extraction and presentation
Vanadium (V) oxide trifluoride can be obtained by reacting vanadium (III) fluoride with oxygen .
It is also produced by hydrolysis of vanadium (V) fluoride .
properties
Vanadium (V) oxide trifluoride is a light yellow moisture-sensitive solid. It has an orthorhombic crystal structure with the space group Pnma (space group no. 62) . With water it hydrolyzes to vanadium (V) oxide hydrate.
use
Vanadium (V) oxide trifluoride is used in organic synthesis for the oxidative coupling of phenol rings , for example for the production of vancomycin .
Individual evidence
- ↑ a b c d e f data sheet Vanadium (V) trifluoride oxide, 99% from AlfaAesar, accessed on October 23, 2013 ( PDF )(JavaScript required) .
- ↑ a b Roger Blachnik (Ed.): Pocket book for chemists and physicists . Volume III: Elements, Inorganic Compounds and Materials, Minerals . founded by Jean d'Ans, Ellen Lax. 4th, revised and revised edition. Springer, Berlin 1998, ISBN 3-540-60035-3 , pp. 790 ( limited preview in Google Book search).
- ^ Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis US, 2011, ISBN 978-1-4398-1461-1 , pp. 451 ( limited preview in Google Book search).
- ↑ a b c Gurdeep Raj: Advanced Inorganic Chemistry Vol-1 . 1975, ISBN 81-87224-03-7 , pp. 1204 ( limited preview in Google Book search).
- ↑ Vanasse, Benoit; O'Brien, Michael K. (2001). "Vanadyl Trifluoride". Encyclopedia of Reagents for Organic Synthesis, doi: 10.1002 / 047084289X.rv005 .




