oxygen
| properties | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| General | |||||||||||||||||||||||||||||||
| Name , symbol , atomic number | Oxygen, O, 8 | ||||||||||||||||||||||||||||||
| Element category | Non-metals | ||||||||||||||||||||||||||||||
| Group , period , block | 16 , 2 , p | ||||||||||||||||||||||||||||||
| Appearance | Colorless gas | ||||||||||||||||||||||||||||||
| CAS number | 7782-44-7 | ||||||||||||||||||||||||||||||
| EC number | 231-956-9 | ||||||||||||||||||||||||||||||
| ECHA InfoCard | 100.029.051 | ||||||||||||||||||||||||||||||
| ATC code | |||||||||||||||||||||||||||||||
| Mass fraction of the earth's envelope | 49.4% | ||||||||||||||||||||||||||||||
| Atomic | |||||||||||||||||||||||||||||||
| Atomic mass | 15.999 (15.99903-15.99977) u | ||||||||||||||||||||||||||||||
| Atomic radius (calculated) | 60 (48) pm | ||||||||||||||||||||||||||||||
| Covalent radius | 73 pm | ||||||||||||||||||||||||||||||
| Van der Waals radius | 152 pm | ||||||||||||||||||||||||||||||
| Electron configuration | [ He ] 2 s 2 2 p 4 | ||||||||||||||||||||||||||||||
| 1. Ionization energy | 13.618 055 (7) eV ≈ 1 313.94 kJ / mol | ||||||||||||||||||||||||||||||
| 2. Ionization energy | 35.12112 (6) eV ≈ 3 388.67 kJ / mol | ||||||||||||||||||||||||||||||
| 3. Ionization energy | 54.93554 (12) eV ≈ 5 300.47 kJ / mol | ||||||||||||||||||||||||||||||
| 4. Ionization energy | 77.41350 (25) eV ≈ 7 469.27 kJ / mol | ||||||||||||||||||||||||||||||
| 5. Ionization energy | 113.8990 (5) eV ≈ 10 989.6 kJ / mol | ||||||||||||||||||||||||||||||
| 6. Ionization energy | 138.1189 (21) eV ≈ 13 326.4 kJ / mol | ||||||||||||||||||||||||||||||
| Physically | |||||||||||||||||||||||||||||||
| Physical state | gaseous (O 2 ) | ||||||||||||||||||||||||||||||
| Crystal structure | cubic | ||||||||||||||||||||||||||||||
| density | 1.429 kg m −3 at 273.15 K. | ||||||||||||||||||||||||||||||
| magnetism | paramagnetic ( Χ m = 1.9 10 −6 ) | ||||||||||||||||||||||||||||||
| Melting point | 54.8 K (−218.3 ° C) | ||||||||||||||||||||||||||||||
| boiling point | 90.15 K (−183 ° C) | ||||||||||||||||||||||||||||||
| Heat of evaporation | 6.82 kJ / mol | ||||||||||||||||||||||||||||||
| Heat of fusion | 0.222 kJ mol −1 | ||||||||||||||||||||||||||||||
| Speed of sound | 317.5 m s −1 at 293 K | ||||||||||||||||||||||||||||||
| Specific heat capacity | 920 J kg −1 K −1 at 298 K | ||||||||||||||||||||||||||||||
| Thermal conductivity | 0.02658 W m −1 K −1 | ||||||||||||||||||||||||||||||
| Chemically | |||||||||||||||||||||||||||||||
| Oxidation states | −2 , −1, 0, +1, +2 | ||||||||||||||||||||||||||||||
| Normal potential | 1.23 | ||||||||||||||||||||||||||||||
| Electronegativity | 3.44 ( Pauling scale ) | ||||||||||||||||||||||||||||||
| Isotopes | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| For other isotopes see list of isotopes | |||||||||||||||||||||||||||||||
| NMR properties | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| safety instructions | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . |
|||||||||||||||||||||||||||||||
Oxygen (also called Oxygenium ; from ancient Greek ὀξύς 'oxys' "sharp, pointed, sour" and γεννάω 'gen-' "generate, give birth", together "acid generator") is a chemical element with the element symbol O. According to its atomic number 8 it is in eighth position in the periodic table and there together with the elements sulfur , selenium , tellurium , polonium and livermorium , which form the chalcogens , in the sixth main group , or 16th IUPAC group . In terms of mass, oxygen is the most common element of the earth's crust with 48.9% , the most common element of the earth's shell and with around 30% the second most common element on earth as a whole ( iron is in 1st place).
Under normal conditions, oxygen occurs in the form of a covalent homodimer , i.e. a compound made up of two oxygen atoms and with the empirical formula O 2 , referred to as molecular oxygen , dioxygen or dioxygen . It is a colorless and odorless gas that is 20.942% in the air . It is involved in many combustion and corrosion processes. Almost all living things need oxygen to live (but plants usually give off more oxygen during photosynthesis than they use). You can usually get it by breathing from the air or by resorption from water (dissolved oxygen). In contrast, in high concentrations it is poisonous to most living things .
The metastable, high-energy and reactive allotropic form made up of three oxygen atoms (O 3 ) is called ozone .
Atomic oxygen , i.e. oxygen in the form of free, individual oxygen atoms, only occurs stably under extreme conditions, for example in the vacuum of space or in hot stellar atmospheres. However, it is of major importance as a reactive intermediate in many reactions in atmospheric chemistry .
history
In the 18th century, Carl Wilhelm Scheele (between 1771 and 1773, published 1777) in Sweden and Joseph Priestley in England (1771, publicly described 1774) discovered oxygen independently of one another in connection with research into combustion processes. Pierre Bayen also came very close to the discovery in 1774.
From the Stone Age to beyond the Middle Ages, fire was an apparition for humans that was accepted as a gift from heaven. Various ideas about the nature of fire arose from the natural philosophers from antiquity to the alchemists . The fire was understood as a basic material of the four-element-doctrine . In the 17th century the idea of a "light, mysterious material" arose. This phlogiston would escape from the burning substance, heat was understood as substance. The German-Swedish pharmacist Carl Wilhelm Scheele made experiments. When brownstone ( manganese dioxide ) or potassium permanganate was heated with concentrated sulfuric acid ( vitriol ), it produced a colorless gas. This gas promoted combustion and Scheele called it "fire air" or "Vitriolluft" after its origin. He found that air is made up of this oxygen and "corrupt air". Two years later, the Englishman Joseph Priestley was able to produce oxygen gas completely independently by heating mercury oxide . The Briton published his findings in 1774, but Scheele did not publish his book Chemische Abhandlung von der Luft und dem Feuer until 1777.
With the discovery of oxygen, its role in combustion was not yet clear. During his experiments, the Frenchman Antoine Lavoisier found that when burned, it was not phlogiston that escaped, but that oxygen was bound. By weighing he proved that a substance was not lighter but heavier after it was burned. The cause was the additional weight of the oxygen absorbed during the combustion process. Initially, oxygen was assumed to be the basic component for the formation of acids . That is why the name Oxygenium ( acid generator ) was proposed by Lavoisier for oxygen in 1779. In fact, most contain inorganic acids in the solution of non-metal oxides in water oxygen. The halogens , such as chlorine and bromine , were therefore long thought to be oxides of unknown elements. Only later was it recognized that hydrogen is responsible for the acidic character ( Humphry Davy , from 1808). In 1883 Karol Olszewski and Zygmunt Wróblewski succeeded for the first time in producing liquid oxygen .
Occurrence
Occurrence on earth
Oxygen is the most abundant and widespread element on earth. It occurs in the earth's atmosphere as well as in the lithosphere , hydrosphere, and biosphere . Oxygen has a mass fraction of 50.5% in the earth's shell (up to 16 km depth, including hydro- and atmosphere). Its mass fraction in air is 23.16% ( volume fraction : 20.95%), in water 88.8% (in seawater, however, only 86%, since larger amounts of non-oxygen-containing salts, e.g. sodium chloride, are dissolved there).
Most of the time, oxygen occurs in its compounds on and in the earth. In addition to water, almost all minerals and therefore rocks in the earth's shell contain oxygen. The most important oxygen-containing minerals include silicates such as feldspars , mica and olivine , carbonates such as calcium carbonate in limestone and oxides such as silicon dioxide as quartz .
In the elementary state, oxygen is in the form of O 2 in gaseous form in the atmosphere and dissolved in water. The amount of the relatively reactive elementary oxygen only remains constant in the long term because oxygen-producing plants supply as much as is used up again by aerobically breathing organisms and other combustion processes. Without this biological cycle, oxygen would only exist in compounds, so elemental oxygen exists in a steady state . The development of the oxygen concentration in the earth's atmosphere is described in the article Development of the earth's atmosphere . The oxygen allotrope O 3 ozone is only present in low concentrations in the atmosphere.
Occurrence in space
In space , oxygen is the third most abundant element after hydrogen and helium . The mass fraction of oxygen in the solar system is around 0.8% (this corresponds to an atomic number of around 500 ppm ).
Oxygen was not created in primordial nucleosynthesis , but it is produced in relatively large quantities in giant stars by burning helium . First, 12 C is formed from three helium nuclei (three- alpha process), which then fuses with another helium nucleus to 16 O. 18 O is formed by fusing a 4 He with a 14 N nucleus. Oxygen also plays a role in generating energy in so-called main sequence stars like the sun. In the CNO cycle (Bethe-Weizsäcker cycle), oxygen is an intermediate product of the nuclear reaction, in which a 4 He nucleus (alpha particle) is created through the capture of protons by a 12 C nucleus that acts as a catalyst . In extremely heavy stars, in the late phase of their development, oxygen burns , in which the oxygen serves as nuclear fuel for reactions that lead to the construction of even heavier nuclei.
Most of the white dwarfs , which according to the current theory represent the final state of 97% of all stars, consist to a large extent of oxygen in addition to helium and carbon.
Extraction and presentation
Technically today, oxygen is obtained almost exclusively by the rectification of air . The process was first developed by Carl von Linde in 1902 ( Linde process ) and made economically viable by Georges Claude . Technically insignificant amounts result as a by-product in the production of hydrogen by electrolysis of water.
To obtain oxygen using the Claude method, air is compressed to 5–6 bar with the help of compressors , cooled and then first freed of carbon dioxide , humidity and other gases through filters . The compressed air is cooled down to a temperature close to the boiling point by gases flowing by from the process. Then it is expanded in turbines. Part of the energy used for compression can be recovered. This makes the process - in contrast to the Linde process , in which no energy is recovered - significantly more economical.
The actual separation of nitrogen and oxygen takes place by distillation in two rectification columns with different pressures. The distillation takes place according to the countercurrent principle, i.e. gas evaporated by the heat of condensation flows upwards, condensed liquid drips downwards. Since oxygen has a higher boiling point than nitrogen, it condenses more easily and thus collects at the bottom, nitrogen at the top of the column. The separation takes place initially at 5–6 bar in the so-called medium pressure column. The resulting oxygen-enriched liquid is then further separated in the low-pressure column (pressure about 0.5 bar). Gaseous nitrogen is passed through the liquid oxygen of the low pressure column. This liquefies and heats the liquid with the heat of condensation given off. The more volatile nitrogen is given off preferentially and purified liquid oxygen remains. This still contains the noble gases krypton and xenon , which are separated in a separate column.
To produce smaller amounts of oxygen, oxygen from the air can be separated from other gases by adsorption . To do this, air flows through molecular sieves . Nitrogen and carbon dioxide are adsorbed and only oxygen and argon get through. This is used in the oxygen concentrator, which is mainly used for medical purposes.
An older process is the barium oxide process based on chemical reactions. It is uneconomical due to the high expenditure of energy. To do this, barium oxide is heated to 500 ° C with a supply of air, during which barium peroxide is formed. When heated to 700 ° C, the previously absorbed oxygen is released again by thermolysis . Before the Linde process was developed, this process was the only way to produce pure oxygen.
-
- Formation of barium peroxide
-
- Release of oxygen and recovery of the barium oxide
Some oxygen-rich inorganic compounds such as potassium permanganate , potassium nitrate (saltpeter), potassium chlorate and potassium chromate give off oxygen when heated or when they react with reducing agents.
Another way to generate oxygen in the laboratory is to decompose hydrogen peroxide on platinum- coated nickel foil .
Pure oxygen can be obtained by electrolysis of 30% potassium hydroxide on nickel electrodes . Hydrogen and oxygen are produced separately from one another.
properties
Physical Properties
Molecular oxygen is a colorless, odorless and tasteless gas that condenses to a bluish liquid at 90.15 K (−183 ° C). In thick layers, gaseous and liquid oxygen show a blue color. Below 54.4 K (−218.75 ° C) oxygen solidifies to form blue crystals. In the solid there are paramagnetic O 2 molecules with an O – O distance of 121 pm (double bond). The element comes in several modifications . Between 54.4 K and 43.8 K, oxygen is in the cubic γ-modification and between 43.8 K and 23.9 K in a rhombohedral β-modification. Finally, below 23.9 K, the monoclinic α-modification is most stable. In contrast to other non-metals, it is paramagnetic and has a diradical character. The triple point is 54.36 K (−218.79 ° C) and 0.1480 kPa. The critical point is at a pressure of 50.4 bar and a temperature of 154.7 K (−118.4 ° C). The critical density is 0.436 g / cm 3 . The density as a liquid at normal pressure is 1.142 g / cm 3 .
Oxygen is not very soluble in water. The solubility depends on the pressure and the temperature. It increases with decreasing temperature and increasing pressure. At 0 ° C, 14.16 mg / l oxygen are dissolved in equilibrium from air under normal pressure (oxygen partial pressure of 212 hPa).

In the oxygen gas discharge spectral tube, the molecular orbitals of oxygen are excited to glow. The operating conditions are a pressure of approx. 5-10 mbar, a high voltage of 1.8 kV, a current of 18 mA and a frequency of 35 kHz. When the ionized gas molecules recombine , the characteristic color spectrum is emitted . Here, ozone is reversibly formed due to the supply of energy.
Molecular orbitals
The bond and the properties of the oxygen molecule can be explained very well with the molecular orbital model . The s and p atomic orbitals of the individual atoms are combined to form binding and antibonding molecular orbitals. The 1s and 2s orbitals of the oxygen atoms become σ s and σ s * - bonding and antibonding molecular orbitals , respectively . Since these orbitals are completely filled with electrons , they do not contribute to the bond. The 2p orbitals become a total of six molecular orbitals with different energy levels. These are the binding σ p -, π x - and π y -, as well as the corresponding antibonding σ p *, π x * and π y * molecular orbitals.
The π orbitals have the same energy. If electrons are distributed in the molecular orbitals, the following distribution of the eight p electrons occurs: six fill the bonding and two in the antibonding π * orbitals; the bond order is therefore (6–2) / 2 = 2. As valence electrons, these two determine the properties of the O 2 molecule. Oxygen has a total of three permitted and energetically achievable quantum mechanical states for the distribution of these electrons .
In the ground state , the spins of the two valence electrons are arranged in parallel, obeying Hund's rule . It is therefore a triplet state with the term symbol 3 Σ g . It is the lowest energy state. Due to the two unpaired electrons, the two π * orbitals are only half occupied. This causes some characteristic properties, such as the diradical character and the paramagnetism of the oxygen molecule.
Despite the formal bond order “two”, no corresponding correct valence line formula can be given for O 2 . <O = O> expresses the double bond character, but ignores both the occupied antibonding orbitals and the radical character. The notation · O̲̅-O̲̅ · is used to emphasize the biradical properties, but only indicates a bond order of one. To indicate bond order two and radical character, the representation <O ÷ O> with radical points on the bond line should be used.
Singlet oxygen
Oxygen has two different excited states , both of which have a significantly higher energy than the ground state. In both states, the spins of the electrons are anti-parallel, contrary to Hund's rule. The more stable excited oxygen is also called singlet oxygen ( 1 O 2 ) after the quantum mechanical term for this state . The two singlet states differ in whether the two electrons are in one (term symbol: 1 Δ g ) or both π * orbitals (term symbol: 1 Σ g ). The 1 Σ g state is energetically less favorable and changes very quickly to the 1 Δ g state. The 1 Σ g state is diamagnetic, but the energetically more stable 1 Δ g state shows due to the existing orbital moment (the quantum number corresponding to the projection of the orbital angular momentum onto the core-core connecting axis - symbolized by Σ, Π, Δ etc. in the 1 Δ g state the value ± 2) Paramagnetism of comparable strength to that of triplet oxygen.
Singlet oxygen can be formed in various ways: both photochemically from triplet oxygen and chemically from other oxygen compounds. A direct extraction from triplet oxygen by irradiation with electromagnetic radiation (e.g. light ) is, however, excluded for quantum mechanical reasons in the form of the selection rules for the emission or absorption of electromagnetic radiation. One way to circumvent this prohibition is to simultaneously irradiate photons and collide two molecules. This unlikely process, which is more likely in the liquid phase, results in the blue color of liquid oxygen ( absorption in the red spectral range). Singlet oxygen can also be represented photochemically with the help of suitable dyes such as methylene blue or eosin . It is obtained chemically from peroxides . When hydrogen peroxide is reacted with sodium hypochlorite , the unstable peroxohypochlorous acid is initially formed, which quickly breaks down into hydrogen chloride or chloride and singlet oxygen. Experimentally, chlorine can also be introduced into an alkaline hydrogen peroxide solution, which then initially produces hypochlorite, which then reacts further. The singlet oxygen reacts quickly with emissions in the red range at 633.4 nm and 703.2 nm to form triplet oxygen.
This form of oxygen is a powerful and selective oxidizing agent and is widely used in organic chemistry . In contrast to normal oxygen, it reacts with 1,3- dienes in a [4 + 2] cycloaddition to form peroxides . Singlet oxygen reacts with alkenes and alkynes in a [2 + 2] cycloaddition.
Chemical properties
Oxygen reacts directly with most other elements to form oxides . There are some exceptions, especially among the non-metals and precious metals . Oxygen only reacts with some elements , for example carbon and sulfur , at high temperatures . A lot of energy is released during combustion and flames develop .
- Burning carbon to carbon dioxide
- Burning sulfur to sulfur dioxide
Reactions with nitrogen are only possible under special conditions, such as lightning , but also in internal combustion engines . This creates nitrogen oxides . Fluorine only forms the unstable compound dioxygen difluoride (O 2 F 2 ) at low temperatures with electrical discharges .
- Oxidation of fluorine to dioxygen difluoride
The noblest metal gold , the halogens chlorine , bromine and iodine , as well as the noble gases do not react directly with oxygen. Some other precious metals such as platinum and silver react poorly with oxygen.
Elementary , gaseous oxygen is relatively inert , many reactions do not take place at all or only slowly under normal conditions . The reason for this is that the reactions with other substances are kinetically inhibited. Either a high activation energy or very reactive radicals are required for the reaction . This barrier can be exceeded by increasing the temperature, light or catalysts such as platinum . In addition, the reaction of many metals is prevented by the fact that the material is coated with a thin metal oxide layer and is thereby passivated . In some reactions, such as the oxyhydrogen reaction , a few radicals are sufficient for a reaction , as they continue to react according to a chain reaction mechanism.
- Combustion of hydrogen into water
Despite the low temperatures, liquid oxygen has a much stronger oxidizing effect than gaseous oxygen. In this, the reactive singlet oxygen forms easily. Even in the presence of water or steam , many oxidations with oxygen proceed more easily.
Reactions with oxygen are almost always redox reactions , in which oxygen usually picks up two electrons and is thus reduced to an oxide . The element is therefore one of the oxidizing agents . Often these reactions take place due to the large binding or lattice energy released with strong heat emission. There is also an explosive running reactions , such as the hydrogen-oxygen reaction or dust explosions of finely divided materials in air or pure oxygen.
Allotropes
In addition to the dioxygen O 2 described in this article, oxygen forms several allotropes that can be differentiated according to the number of oxygen atoms. The most important allotrope is ozone O 3 , next to the rarer allotropes Tetra oxygen (O are 4 ) and Oktasauerstoff (O 8 known).
ozone
Ozone (O 3 ) is a blue gas with a characteristic smell that consists of three oxygen atoms. It is unstable, very reactive, and a strong oxidizer. It is formed from molecular oxygen and oxygen atoms, but also z. B. by reaction of nitrogen dioxide with oxygen under UV radiation .
Due to its high reactivity, it is rather detrimental to human health near the ground - in the ozone layer of the earth's atmosphere, on the other hand, ozone plays an important role in the absorption of the UV radiation hitting the earth.
Other allotropes
A high pressure phase of the oxygen arises as a red solid at pressures greater than 10 GPa . According to crystallographic studies, it is assumed that these are octase oxygen O 8 rings. In addition, tetrasoxy exists as a very rare and unstable allotrope of oxygen. It could be detected in the mass spectrometer in 2001. It occurs in low concentrations in liquid oxygen.
Isotopes
The most common stable oxygen isotopic is 16 O (99.76%), 18 O (0.20%) and 17 O (0.037%) also occur. In addition to the stable oxygen isotopes, a total of 13 unstable, radioactive nuclides from 12 O to 28 O are known that can only be produced artificially. Their half-lives are usually only milliseconds to seconds, 15 O has the longest half-life of two minutes and is often used in positron emission tomography .
As the only stable isotope of the rare has 17 O a nuclear spin of 5 / 2 , and can thus for NMR are used -Untersuchungen. The other stable isotopes have nuclear spin 0 and are therefore NMR-inactive.
Indirect temperature measurement via the δ 18 O signal
Water molecules with 16 O, which is 12% lighter, evaporate faster. Therefore, ice layers with a higher relative proportion of 18 O must originate from warmer times, since 18 O water only contributes to the formation of clouds during the strong evaporation of warmer periods . The higher the global temperature, the further clouds laden with heavy oxygen isotopes can penetrate into the polar regions without first raining.
In colder periods there is more 18 O in marine sediments . Sea ice consists mainly of the lighter water molecules from 16 O. If there is a strong formation of new sea ice in a cold phase, more sea water from 18 O remains , which is reinforced in sediment layers by the permanent storage of oxygen in the calcareous shells of sea animals ( calcium carbonate ) this time is detectable. There are also regional differences in the 18 O accumulation in organisms according to the type of their drinking water source.
An isotope analysis of ice cores or sediment samples and the determination of the 18 O / 16 O ratio with the help of a mass spectrometer provide information about the average temperature and thus global warming and cooling in earlier times. In addition, by determining the number of oscillations between warm (summer) and cold (winter), the age of the drill core can be determined exactly.
use
Oxygen is used for industrial combustion, oxidation and heating processes, in medicine and in aerospace.
medicine
Oxygen for use in human medicine is subject to strict controls due to legal regulations. The medical oxygen filled in bottles marked in white is considered a finished medicinal product in Germany within the meaning of the Medicines Act (AMG).
Caution is advised in the administration of oxygen when the patient (see from a chronic lung disease COPD ) with increased CO 2 - partial pressure suffer. With them, the sudden “oversupply” of oxygen can lead to CO 2 anesthesia with respiratory arrest.
Emergency medicine
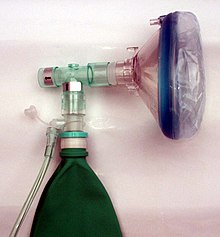
Injuries and many diseases of the lungs as well as some heart diseases and especially shock states can lead to a lack of oxygen ( hypoxia ) in the arteries and in the tissues of vital organs. For this reason, patients in emergency and intensive care medicine are very often given supplemental oxygen. In the case of independently breathing patients, the ambient air is enriched with oxygen with the help of various probes and masks; in the case of artificially ventilated patients, the oxygen is added in the ventilator . The effect of oxygen enrichment in the blood can be measured with the help of pulse oximetry or with the help of blood gas analyzes .
Long-term oxygen therapy
In diseases with severe chronic oxygen deficiency in the blood, a long-term supply of oxygen for several hours every day ( long-term oxygen therapy ) improves both quality of life and survival time. The pure oxygen can lead to problems during ventilation due to the displacement of carbon dioxide from the vessels and to an undesirable increase in brain activity in the hypothalamus , the insula and the hippocampus . These negative consequences are avoided by adding carbon dioxide.
Cluster headache
According to the recommendations of the World Health Organization , breathing oxygen is suitable for the treatment of cluster headache attacks. The use of highly concentrated oxygen by means of special mask systems usually effectively relieves the symptoms within minutes.
technology
Oxygen is used industrially mainly in metallurgy for the production of pig iron and steel, as well as in copper refining. Pure oxygen or oxygen-enriched air is used here on the one hand to achieve high temperatures and on the other hand to refine the crude steel, i.e. H. to remove unwanted additions of carbon , silicon , manganese and phosphorus , which are oxidized and separated. Compared to air, pure oxygen has the advantage that no nitrogen is introduced into the melt. Nitrogen has a negative influence on the mechanical properties of steel (see also Thomas method ). In chemical processes, oxygen is mostly used for the oxidation of various raw materials, such as the olefin oxidation of ethene to ethylene oxide and the partial (partial) oxidation of heavy oil and coal. Oxygen is also required for the production of hydrogen and synthesis gas and the production of sulfuric and nitric acid . Other important products made by oxidation with oxygen are acetylene (ethyne), acetaldehyde , acetic acid , vinyl acetate and chlorine .
Different fuel gases ( propane , hydrogen , ethyne, etc.) only achieve sufficiently hot and soot-free flames for welding and brazing or melting and making glass malleable by mixing with oxygen . After heating up and igniting, concrete is cut with a (self-burning) oxygen lance or iron is flame cutting with a sharp oxygen jet alone.
Oxygen is also used to produce ozone , as an oxidizing agent in fuel cells and in semiconductor technology. In rocket technology , liquid oxygen is used as an oxidizing agent and is abbreviated to LOX (liquid oxygen) .
In environmental technology , wastewater is freed from organic pollutants and toxins more quickly by the introduction of oxygen gas by bacteria. In many drinking water works, drinking water treatment with ozone ( ozonation ) serves to oxidize organic substances or iron, for example, and to disinfect and can lead to a significantly better cleaning effect of the filter systems.
For food technology , oxygen is approved as a food additive as E 948 and is used - in addition to nitrogen, carbon dioxide and nitrous oxide as propellant gas, packing gas, gas for whipping cream (whipped cream) and the like. used.
Wellness
In the wellness and food industries, products that are enriched with oxygen are occasionally advertised. For example, packaged water is sold that is said to have a higher oxygen content. A positive effect on health and well-being is not to be expected, because oxygen only dissolves in small amounts in water and is absorbed in many orders of magnitude more - namely with every breath - via the lungs than via the stomach.
Battery technology
The zinc-air cell is a primary element that takes in air from the surrounding atmosphere and chemically converts the oxygen with the metal zinc to generate electrical voltage and current. The advantage of this type of battery is that it can deliver its relatively high energy density (with low power consumption) over an extended period of time. Keeping the ventilation opening closed with an adhesive strip (seal) before use helps to reduce the aging of the cell during storage. Fist-sized cuboid cells of this type in a plastic housing were used around 1980 in flashing lights to secure construction sites and are still common today as button cells for behind-the-ear hearing aids .
Research and development are also moving in the direction of metal-oxygen batteries based on alkali metals. The aim is to exceed the energy density (per mass) of lithium-ion batteries .
Biological importance

In nature, oxygen is in a constant cycle . It is continuously released from water by autotrophic organisms such as cyanobacteria ("blue-green algae"), algae and plants during oxygenic photosynthesis by photolysis . It is an end product of this biochemical reaction and is released into the environment. Cyanobacteria were probably the first organisms to accumulate molecular oxygen as a waste product in the atmosphere . Before that, there was a practically oxygen-free, anaerobic atmosphere on Earth .
- simplified net reaction equation for oxygenic photosynthesis
Most aerobic organisms , including most eukaryotes , including humans and plants , and many bacteria , need this oxygen to live . Eukaryotes need it to generate energy through oxidation in the mitochondria . The oxygen is reduced to water again in the respiratory chain . The oxygenation of metabolites using enzymes ( oxygenases ) is often the degradation of materials used. The reaction requires oxygen and takes place in all aerobic living things .
- simplified net reaction equation for aerobic respiration
Since oxygen and some of its compounds are very reactive and can destroy cell structures, organisms have protective enzymes such as catalase and peroxidase . For organisms , which these enzymes are missing, oxygen is toxic . When reduction of the oxygen produced reactive oxygen species such as free radicals , which are also biological molecules can destroy. If they are not intercepted quickly enough, so-called oxidative stress occurs , which is made responsible for aging processes .
In the phagocytes (scavenger cells) of the immune system , these reactive oxygen species ( hydrogen peroxide and hyperoxide ions ) serve in addition to enzymes to destroy ingested pathogens .
Problematic effects
If pure oxygen or air with a higher proportion of oxygen is inhaled over a long period of time, it can poison the lungs, the so-called Lorrain-Smith effect . The alveoli of the lungs are prevented from swelling in their normal function.
The Paul Bert effect describes oxygen poisoning of the central nervous system . This can occur with high pressure breathing of any oxygen-nitrogen mixture, but the risk increases as the oxygen content and total pressure increase. At partial oxygen pressures above 1.6 bar, poisoning occurs within a relatively short time . This plays a role in diving , for example , as it limits the maximum diving depth depending on the oxygen partial pressure.
In space travel , for example, pure oxygen is breathed in spacesuits , albeit under greatly reduced pressure in order to minimize health consequences and because the spacesuit would become too stiff under normal pressure.
Hyperoxide anions in metabolism
Hyperoxide anions (old name: superoxide anions) are simply negatively charged and radical oxygen ions (O 2 - ) that are created by electron transfer to molecular oxygen. These are extremely reactive. Sometimes they are formed as a by-product of metabolism (metabolism) such as by side reactions in some oxidases (xanthine oxidase). Hyperoxide anions are also formed in photosynthesis complex I and as a by-product of the respiratory chain ( mitochondrial respiration ). Xenobiotics and cytostatic antibiotics promote their development. When inflammation occurs , a membrane-based NADPH -dependent oxidase releases hyperoxide anions into the extracellular environment. They lead to oxidative stress . For example, when fatty acids are broken down in the peroxisomes, electrons are transferred from FADH 2 to molecular oxygen. The resulting hyperoxide anions can react further to form the cell poison hydrogen peroxide . During the respiratory chain , these radical oxygen species are produced in small quantities. There are suspicions that the genetic damage caused by such oxygen species is involved in aging processes. It is therefore for the organism to degrade rapidly this Hyperoxidanionen essential. This is done using superoxide dismutase .
Analytics
Classic analytics
Dissolved oxygen oxidizes divalent manganese to higher oxidation states. According to the Winkler method, this is again completely reduced by iodide . The amount of substance of the iodine formed is in a stoichiometric ratio of 1: 2 to the amount of substance of the originally dissolved oxygen and can be back-titrated iodometrically with thiosulfate .
The glow chip test is used as a simple proof of the presence of oxygen .
Instrumental quantitative analysis
The lambda probe used to control the combustion of gasoline engines measures the oxygen content in the car exhaust in relation to the O 2 content in the ambient air. For this purpose, the exhaust gas flow is passed through a yttrium- doped zirconium dioxide tube , which is provided with platinum electrodes on the inside and outside . The outer electrode is in contact with the ambient air. Different O 2 partial pressures at the electrodes lead to an electrical potential difference, which is measured. The advantages of this measurement technology are the low detection limit of a few ppm and the large operating temperature range (300 ° C to 1500 ° C).
The Clark electrode is an amperometric sensor for the electrochemical determination of dissolved, gaseous oxygen. Platinum and Ag / AgCl reference electrodes are located in an electrolyte system which is separated from the sample solution by a gas-permeable Teflon membrane. Dissolved oxygen can diffuse through the membrane into the electrolyte solution and is cathodically reduced in a potential range of −600 mV to −800 mV. The measured current is proportional to the oxygen concentration in the sample solution.
The optical methods make use of the fact that oxygen can quench the fluorescence of excited molecules. On the basis of fluorescent transition metal complexes, so-called optrodes have been developed which determine the oxygen content via the fluorescence quenching of probe molecules. Metal-ligand complexes are often used as probe molecules. Ru (II), Ir (II), Pt (II) and Pd (II) have proven themselves as metal ions, and various bipyridines , phenanthrolines and (fluorinated) porphyrins as ligands . The probes are embedded in polymer matrices. The excitation takes place mostly with LEDs or laser diodes . A distinction is made between point measurements z. B. using optical fiber optics and imaging measurement methods using planar sensor films. Detection limits of 5 ppbv (O 2 , ≈ 5.1 · 10 −6 hPa) could be achieved with optrodes , which corresponds to a concentration in water of 7 pM.
links
Oxygen forms compounds with almost all elements - with the exception of the noble gases helium , neon and argon . Since oxygen is very electronegative , it occurs in almost all of its compounds in the −II oxidation state , only in peroxides −I. These ions are also known as closed-shell ions. Peroxides are usually unstable and easily convert to oxides .
Oxygen only has positive oxidation numbers in compounds with the even more electronegative element fluorine , with which it forms compounds with the oxidation state + I (dioxygen difluoride O 2 F 2 ) and + II ( oxygen difluoride OF 2 ). Since they have negative polarization with fluorine, they are not referred to as oxides, but as fluorides , more precisely oxygen fluorides .
In addition to the oxide compounds, oxygen occurs in ionic compounds and radicals as peroxide - (O 2 2− ), hyperoxide (O 2 - (oxidation state −1/2)) and ozonide anion (O 3 - (oxidation state −1/3)) as well as a dioxygenyl cation (O 2 + ).
Depending on the binding partner, oxygen forms both ionic and covalent compounds.
Inorganic oxygen compounds
The inorganic oxygen compounds include the compounds of oxygen with metals , semi-metals , non-metals such as hydrogen , carbon , nitrogen , sulfur and the halogens . They are among the most important connections of all.
Oxides
Most oxygen compounds are oxides . In them, the oxygen, bound ionically or covalently , occurs in the −II oxidation state . Many naturally occurring salts , which are often important sources in the manufacture of metals , are oxides.
With the metals, oxygen forms ionically structured and usually basic oxides in low oxidation states.
- When calcium is burned in the presence of oxygen, calcium oxide is formed .
- Calcium hydroxide is formed when calcium oxide reacts with water .
As the oxidation level increases, the oxides increasingly have amphoteric ( zinc (II) oxide , aluminum (III) oxide ) and finally acidic character ( chromium (VI) oxide ).
- Chromium (VI) oxide reacts with water to form chromic acid
Oxygen only forms covalent oxides with non-metals . The oxides of non-metals in low oxidation states mostly react neutrally ( nitrous oxide ), and with increasing oxidation state they react increasingly acidic.

Among the oxygen compounds of the non-metals, those with hydrogen play a special role. Oxygen forms two compounds with hydrogen. First of all there is water , without which there would be no life on earth. The second compound is hydrogen peroxide (H 2 O 2 ), a thermodynamically unstable compound that is used as an oxidizing and bleaching agent .
Although most oxygen-containing carbon compounds are classified in the field of organic chemistry, there are a few important exceptions. The simple oxides of carbon, carbon monoxide (CO) and carbon dioxide (CO 2 ), as well as carbonic acid and its salts, the carbonates , are regarded as inorganic compounds.
If lower amounts of oxide ions are known in a salt-like compound than are to be expected based on the stoichiometry and valence of the oxygen, one speaks of suboxides . Element-element bonds occur in these, the formal oxidation state of the element is below +1. Elements that form suboxides are the alkali metals rubidium and cesium , but also boron and carbon .
Oxygen compounds with oxygen-oxygen bonds
Oxygen forms compounds with oxygen-oxygen bonds, especially with alkali metals. These include peroxides , hyperoxides and ozonides . Peroxides such as hydrogen peroxide have the O 2 2− ion and a formal oxygen oxidation state of −1. By easily breaking the oxygen-oxygen bond, they easily form radicals that have a bleaching effect on organic substances and are accordingly used as bleaching agents . Organic peroxides are also known.
The radical dioxide (1 -) - anion O 2 - occurs in hyperoxide n , the formal oxidation state is −½ for each oxygen atom. Hyperoxide ions are formed in the metabolism and are among the reactive oxygen species , salt-like hyperoxides are only known from alkali metals apart from lithium. Ozonides are derived from ozone and accordingly have the O 3 - anion. Salt-like ozonides, like hyperoxides, are known from all alkali metals except lithium; there are also organic ozonides, which are formed by the addition of ozone to alkenes .
Hydroxides
Another large group of oxygen compounds are the hydroxides with the participation of hydrogen . These are predominantly ionic compounds, which have the hydroxide ion in common. Except for the hydroxides of the alkali metals such as sodium hydroxide (NaOH) or potassium hydroxide (KOH), they are generally sparingly soluble in water .
Oxo acids
When non-metal oxides and metal oxides of metals in high oxidation states react with water, the so-called oxygen acids are formed , which are responsible for the name given to the oxygen.
- When sulfur trioxide is dissolved in water, sulfuric acid is formed .
The strongest inorganic oxygen acids are derived from the non-metals nitrogen ( nitric acid ) and sulfur (sulfuric acid) as well as the halogens ( halogen oxygen acids ). The rule here is that the acid strength ( pK s value ) increases with the number of oxygen atoms:
| Name of the acid | formula | Name of the salts | Acid constant (pK s ) |
|---|---|---|---|
| carbonic acid | H 2 CO 3 | Carbonates | 6.35 |
| Orthosilicic acid | H 4 SiO 4 | Silicates | 9.51 |
| Cyanic acid | HOCN | Cyanates | 3.7 |
| Isocyanic acid | HNCO | Cyanates | 3.92 |
| Hypo-nitrous acid | H 2 N 2 O 2 | Hyponitrites | 7.21 |
| Nitrous acid | ENT 2 | Nitrites | 3.29 |
| nitric acid | ENT 3 | Nitrates | −1.37 |
| Phosphinic acid | H 3 PO 2 | Phosphinates | 2.0 |
| Phosphonic acid | H 3 PO 3 | Phosphonates | 2.0 |
| phosphoric acid | H 3 PO 4 | Phosphates | 2.16 |
| Thiosulfuric acid | H 2 S 2 O 3 | Thiosulfates | 0.6 |
| sulphurous acid | H 2 SO 3 | Sulfites | 1.81 |
| sulfuric acid | H 2 SO 4 | Sulfates | −3 |
| Hypochlorous acid | HClO | Hypochlorites | 7.54 |
| Chlorous acid | HClO 2 | Chlorites | 1.97 |
| Chloric acid | HClO 3 | Chlorates | −2.7 |
| Perchloric acid | HClO 4 | Perchlorates | −10 |
Organic oxygen compounds
Along with carbon , hydrogen and nitrogen , oxygen is one of the most important elements in organic chemistry . It forms a large number of important functional groups that contain both carbon-oxygen single bonds and - in the carbonyl group - carbon-oxygen double bonds .
One of the simplest organic compounds that contain oxygen is methanal (H 2 CO), which only differs from carbon dioxide (CO 2 ) in that two hydrogen atoms are bonded to the carbon instead of the second oxygen atom. For the classification into organic chemistry, however, it is important that methanal is derived from the organic alcohol methanol (CH 3 OH), which in turn is a derivative of the simplest alkane methane (CH 4 ).
The main compound classes:
- Alcohols : Some important classes of compounds are derived from the carbon-oxygen single bond. The first are the alcohols, in which a carbon and a hydrogen atom ( hydroxyl group ) are bonded to the oxygen atom . The best-known and at the same time simplest representatives of this group are methanol CH 3 OH and ethanol C 2 H 5 OH.
- Phenols : These molecules contain at least one hydroxy group attached to an aromatic ring .
- Ether : If two carbon atoms are bonded to the oxygen atom, the group is called an ether group and the substance class is called ether. A well-known representative of ethers is the important solvent diethyl ether (C 2 H 5 ) 2 O.
- Aldehydes : The carbonyl group is a very versatile functional group that is found in many substance classes. These differ in which additional groups are attached to the carbon atom. The aldehyde group R – CHO, in which a hydrogen atom is bonded to the carbon atom of the carbonyl group, occurs in aldehydes such as acetaldehyde .
- Ketones : They contain the keto group , R – CO – R, in which two hydrocarbon radicals are bonded to the carbon atom of the carbonyl group. One example is acetone .
- Carboxylic acids : The carboxy group R – COOH of the carboxylic acids has both a carbonyl group and a hydroxyl group on one carbon atom. The most important carboxylic acids are formic and acetic acid .
- Ester : The ester group R – CO – O – R ′ has a structure similar to the carboxy group . The proton of the carboxylic acid has been exchanged for another hydrocarbon residue. The esters formed from carboxylic acids and alcohols are named accordingly. An example is ethyl acetate made from acetic acid and ethanol (ethyl alcohol).
- Carboxamides : In them, the hydroxyl group of the carboxy group has been replaced by an amino group .
Another important group of organic oxygen compounds are the carbohydrates or saccharides . Chemically, these are polyhydroxycarbonyl compounds ( hydroxyaldehydes or hydroxyketones ). So they combine properties of alcohols with those of aldehydes and ketones.
There are also a number of other compounds with functional groups in which the oxygen is bound to another heteroatom such as nitrogen , sulfur or phosphorus , for example in organic phosphates (such as ATP or within the DNA molecules ).
See also
literature
- AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , pp. 497-540.
- NN Greenwood, A. Earnshaw: Chemistry of the Elements. 1st edition. VCH Verlagsgesellschaft, Weinheim 1988, ISBN 3-527-26169-9 , pp. 775-839.
- Ralf Steudel: chemistry of non-metals. de Gruyter, Berlin 1998, ISBN 3-11-012322-3 .
- Nick Lane: Oxygen - the molecule that made the world. Oxford Univ. Press, Oxford 2003, ISBN 0-19-860783-0 .
Web links
Individual evidence
- ^ Harry H. Binder: Lexicon of the chemical elements. S. Hirzel Verlag, Stuttgart 1999, ISBN 3-7776-0736-3 .
- ↑ The values for the properties (info box) are taken from www.webelements.com (oxygen) , unless otherwise stated .
- ↑ The standard value recommended by the IUPAC is given, since the isotopic composition of this element can vary locally, the range given in brackets results for the mean atomic mass. See: Michael E. Wieser, Tyler B. Coplen: Atomic weights of the elements 2009 (IUPAC Technical Report). In: Pure and Applied Chemistry. 2010, p. 1, doi: 10.1351 / PAC-REP-10-09-14 .
- ^ IUPAC, Standard Atomic Weights Revised 2013 .
- ↑ a b c d e f Entry on oxygen in Kramida, A., Ralchenko, Yu., Reader, J. and NIST ASD Team (2019): NIST Atomic Spectra Database (ver. 5.7.1) . Ed .: NIST , Gaithersburg, MD. doi : 10.18434 / T4W30F ( https://physics.nist.gov/asd ). Retrieved June 11, 2020.
- ↑ a b c d e f Entry on oxygen at WebElements, https://www.webelements.com , accessed on June 11, 2020.
- ↑ a b c Entry on oxygen in the GESTIS substance database of the IFA , accessed on August 9, 2016(JavaScript required) .
- ↑ Robert C. Weast (Ed.): CRC Handbook of Chemistry and Physics . CRC (Chemical Rubber Publishing Company), Boca Raton 1990, ISBN 0-8493-0470-9 , pp. E-129 to E-145. Values there are based on g / mol and given in cgs units. The value specified here is the SI value calculated from it, without a unit of measure.
- ↑ a b Yiming Zhang, Julian RG Evans, Shoufeng Yang: Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. In: Journal of Chemical & Engineering Data. 56, 2011, pp. 328-337, doi: 10.1021 / je1011086 .
- ↑ Entry on Oxygen in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ Entry on oxygen. In: Römpp Online . Georg Thieme Verlag, accessed on December 20, 2012.
- ^ Wissenschaft-Online-Lexika: Entry on oxygen in the Lexicon of Geosciences, accessed on December 20, 2012.
- ↑ E. Pilgrim: Discovery of the elements. Mundus Verlag, Stuttgart 1950.
- ^ Rudolf Frey , Otto Mayrhofer , with the support of Thomas E. Keys and John S. Lundy: Important data from the history of anesthesia. In: R. Frey, Werner Hügin , O. Mayrhofer (Ed.): Textbook of anesthesiology and resuscitation. Springer, Heidelberg / Basel / Vienna 1955; 2nd, revised and expanded edition. With the collaboration of H. Benzer. Springer-Verlag, Berlin / Heidelberg / New York 1971. ISBN 3-540-05196-1 , pp. 13–16, here: p. 14.
- ↑ Joseph Priestley: An Account of Further Discoveries in Air. By the Rev. Joseph Priestley, LL.DFRS in Letters to Sir John Pringle, Bart. PRS and the Rev. Dr. Price, FRS In: Phil. Trans. 65, Jan. 1, 1775, pp. 384-394; doi: 10.1098 / rstl.1775.0039 ( full text ).
- ^ Claude Allègre, Gérard Manhès, Éric Lewin: Chemical composition of the Earth and the volatility control on planetary genetics. In: Earth and Planetary Science Letters. 185 (1-2), 2001, pp. 49-69; doi: 10.1016 / S0012-821X (00) 00359-9 .
- ↑ a b dtv-Atlas Chemie. Volume 1, dtv-Verlag, 2000.
- ^ A b c d e f A. F. Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , pp. 497-540.
- ↑ AM Davies (Ed.): Treatise on Geochemistry. Volume 1: Meteorites, Comets, and Planets. Elsevier, 2003, ISBN 0-08-044720-1 .
- ↑ Glenn J. MacPherson: Oxygen in the solar system. Mineralogical Society of America, Chantilly 2008, ISBN 978-0-939950-80-5 .
- ↑ R. Kippenhahn, A. Weigert: Stellar Structure and Evolution. 1st edition. Springer, Berlin 1991, ISBN 3-540-58013-1 .
- ↑ a b Functional principle of an oxygen production plant .
- ^ A b c N. N. Greenwood, A. Earnshaw: Chemistry of the elements. 1st edition. VCH Verlagsgesellschaft, Weinheim 1988, ISBN 3-527-26169-9 , pp. 775-839.
- ↑ Principle of gas separation through adsorption (air liquide) .
- ^ MJ Kirschner: Oxygen In: Ullmann's Encyclopedia of Industrial Chemistry . Wiley-VCH Verlag, Weinheim 2012, doi : 10.1002 / 14356007.a18_329
- ^ National Physical Laboratory: Kaye and Laby Tables of Physical and Chemical Constants. 16th edition. 1995; D. Ambrose, MB Ewing, ML McGlashan: Critical constants and second virial coefficients of gases. ( Memento from March 24, 2017 in the Internet Archive )
- ↑ JA Dean: Lange's Handbook of Chemistry. 15th edition. McGraw-Hill, 1999; Section 6; Table 6.5 Critical Properties
- ↑ Physical properties
- ↑ Oxygen spectral tube .
- ↑ Keisuke Hasegawa: Direct measurements of absolute concentration and lifetime of singlet oxygen in the gas phase by electron paramagnetic resonance. In: Chemical Physics Letters. 457 (4-6), 2008, pp. 312-314; doi: 10.1016 / j.cplett.2008.04.031 .
- ↑ NV Shinkarenko, VB Aleskovskiji: Singlet Oxygen: Methods of Preparation and Detection. In: Russian Chemical Reviews. 50, 1981, pp. 320-231; doi: 10.1070 / RC1981v050n03ABEH002587 .
- ↑ Peter Lechtken: Singlet Oxygen . In: Chemistry in Our Time . tape 8 , no. 1 , 1974, p. 11-16 , doi : 10.1002 / ciuz.19740080103 .
- Jump up ↑ Video showing singlet oxygen glowing red
- ^ University of Bayreuth: Carbon dioxide and carbonic acid
- ↑ Catalytic oxidation of sulfur dioxide
- ↑ Juergen Carstens: Calculation of the NOx raw emissions of an internal combustion engine in stratified charge operation . In: The IP.com Journal. 2003.
- ↑ G. Brauer; Handbook of preparative inorganic chemistry, F. Enke Verlag 1978, ISBN 978-3-432-26081-5 .
- ↑ F. Cacace, G. De Petris, A. Troiani: Experimental Detection of Tetraoxygen In: Angewandte Chemie (International ed. In English). Volume 40, Number 21, November 2001, pp. 4062-4065. PMID 12404493 ; doi : 10.1002 / 1521-3773 (20011105) 40:21 <4062 :: AID-ANIE4062> 3.0.CO; 2-X
- ↑ Philip Ball: New form of oxygen found. In: Nature News. November 16, 2001, accessed on September 10, 2013 (English, doi: 10.1038 / news011122-3 ).
- ↑ a b G. Audi, FG Kondev, Meng Wang, WJ Huang, S. Naimi: The NUBASE2016 evaluation of nuclear properties. In: Chinese Physics C. 41, 2017, S. 030001, doi : 10.1088 / 1674-1137 / 41/3/030001 ( full text ).
- ↑ NMR properties of oxygen at www.webelements.com
- ↑ Law on the trade in medicinal products (Drugs Act - AMG), Section 50. Version of the announcement of December 12, 2005 ( Federal Law Gazette 2005 I p. 3394 ), last amended by Article 30 of the law of March 26, 2007.
- ^ A. New: Oxygen: kill or cure? Prehospital hyperoxia in the COPD patient. In: Emerg Med J. 23, 2006, pp. 144-146. PMID 16439751 .
- ^ FJ Andrews, JP Nolan: Critical care in the emergency department: monitoring the critically ill patient. In: Emerg Med J. 23, 2006, pp. 561-564. PMID 16794104 .
- ↑ German Society for Pneumology: Recommendations for long-term oxygen therapy in severe chronic hypoxemia. In: Pneumology. 74, 1993, pp. 2-4.
- ↑ S. Iscoe, JA Fisher: hyperoxia-induced hypocapnia: risk of underappreciated. In: Chest. 128 (1), Jul 2005, pp. 430-433. PMID 16002967 ; doi: 10.1378 / chest.128.1.430 .
- ^ Paul M. Macey, Mary A. Woo, Ronald M. Harper: Hyperoxic Brain Effects Are Normalized by Addition of CO 2 . In: Public Library of Science Medicine. 4 (5), 2007, p. E173; doi: 10.1371 / journal.pmed.0040173 .
- ^ WHO - Lifting The Burden: Aids for management of common headache disorders in primary care. (PDF file; 279 kB).
- ↑ Guideline for cluster headache and trigeminal autonomic headache . Published by the guidelines commission of the German Society for Neurology , the German Migraine and Headache Society , the Austrian Society for Neurology , the Swiss Neurological Society and the Professional Association of German Neurologists . Status: May 14, 2015, valid until May 13, 2020.
- ↑ Dimitriadou Agapi, Komi Evangelia, Lykou Maria: Urban Metabolism - Water Treatment in Chapter 5, Section 5.7
- ↑ Additive Admissions Ordinance : Annex 3 (to Section 5, Paragraph 1 and Section 7) Generally permitted additives .
- ↑ Soft drinks. The inspection offices for food control and animal health, Baden-Württemberg. 2008.
- ↑ Graz: Researchers extend the lifespan of batteries orf.at, March 20, 2017, accessed on March 21, 2017.
- ↑ Nika Mahne, Bettina Schafzahl a. a .: Singlet oxygen generation as a major cause for parasitic reactions during cycling of aprotic lithium-oxygen batteries. In: Nature Energy. 2, 2017, p. 17036, doi : 10.1038 / nenergy.2017.36 .
- ↑ Spectrum of Science Verlagsgesellschaft mbH: Photosynthesis
- ↑ Ulrich Helmich: Breathing, Dissimilation
- ^ LG Anderson, C. Haraldsson, R. Lindgren: Gran linearization of potentiometric Winkler titration. In: Marine Chemistry. 37 (3-4), 1992, pp. 179-190; doi: 10.1016 / 0304-4203 (92) 90077-N .
- ↑ J. Riegel, H. Neumann, H.-M. Wiedenmann: Exhaust gas sensors for automotive emission control. In: Solid State Ionics. 152/153, 2002, pp. 783-800; doi: 10.1016 / S0167-2738 (02) 00329-6 .
- ↑ G. Eranna, BC Joshi, DP Runthala, RP Gupta: Oxide Materials for Development of Integrated Gas Sensors - A Comprehensive Review. In: Crit. Rev. Solid State Mat. Sci. 29, 2004, pp. 111-188; doi: 10.1080 / 10408430490888977 .
- ^ A b E. Gnaiger, H. Forstner: Polarographic Oxygen Sensors. Springer-Verlag, Berlin / Heidelberg / New York 1983.
- ↑ JR Stetter, J. Li: Amperometric Gas Sensors - A Review. In: Chemical Reviews. 108 (2), 2008, pp. 352-366; doi: 10.1021 / cr0681039 .
- ↑ C. McDonagh, CS Burke, BD MacCraith: Optical Chemical Sensors. In: Chemical Reviews. 108 (2), 2008, pp. 400-422; doi: 10.1021 / cr068102g .
- ↑ Y. Amao: Probes and Polymers for Optical Sensing of Oxygen. In: Microchimica Acta . 143, 2003, pp. 1-12; doi: 10.1007 / s00604-003-0037-x .
- ↑ S. Arain, S. Weiss, E. Heinzle, GT John, C. Krause, I. Klimant: Gas Sensing in microplates with Optodes: Influence of Oxygen Exchange Between Sample, Air and plate material. In: Biotechnology and Bioengineering . Vol. 90, No. 3, 2005, pp. 271-280.
- ↑ MIJ Stich, LH Fischer, OS Wolfbeis: Multiple fluorescent chemical sensing and imaging. In: Chemical Society Reviews . 39, 2010, pp. 3102-3114.
- ↑ P. Lehner, C. Staudinger, SM Borisov, I. Klimant: Ultra-sensitive optical oxygen sensors for characterization of nearly anoxic systems. In: Nature Communications. 5, 2014, p. 4460; doi: 10.1038 / ncomms5460 .
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 .