Californium
| properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Generally | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name , symbol , atomic number | Californium, Cf, 98 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Element category | Actinides | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group , period , block | Ac , 7 , f | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Look | silvery metal | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | 7440-71-3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic mass | 251 and | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | 186 ± 2 pm pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 225 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [ Rn ] 5 f 10 7 s 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1. Ionization energy | 6th.28166 (25) eV ≈ 606.09 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2. Ionization energy | 12th.0 (4) eV ≈ 1 160 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3. Ionization energy | 22nd.4 (4) eV ≈ 2 160 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4. Ionization energy | 37.7 (4) eV ≈ 3 640 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5. Ionization energy | 51.9 (1.9) eV ≈ 5 010 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physically | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical state | firmly | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Modifications | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | hexagonal | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| density | 15.1 g / cm 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1173 K (900 ° C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar volume | 16.50 10 −6 m 3 mol −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemically | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | (+2), +3 , (+4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal potential | −1.910 V (Cf 3+ + 3 e - → Cf) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | 1.30 ( Pauling scale ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| For other isotopes, see the list of isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard and safety information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Radioactive |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Californium (seldom also spelled Californium ) is an artificially created chemical element with the element symbol Cf and the atomic number 98. In the periodic table it is in the group of actinides ( 7th period , f-block ) and is also one of the transuranic elements . It was named after the University of California and the US state of California , where it was discovered. Californium is a radioactive metal . It was first created in February 1950 from the lighter element curium . It is produced in small quantities in nuclear reactors . It is mainly used for mobile and portable neutron sources , but also for the generation of higher transuranic elements and transactinides .
history
Just as americium (atomic number 95) and curium (96) were discovered almost simultaneously in 1944 and 1945, the elements berkelium (97) and californium (98) were similarly discovered in 1949 and 1950 .
Californium was produced for the first time on February 9, 1950 at the University of California at Berkeley by Stanley G. Thompson , Kenneth Street, Jr. , Albert Ghiorso and Glenn T. Seaborg by bombarding atomic nuclei of Curium with α-particles . It was the sixth Transuranic that was discovered. The discovery was published at the same time as that of the Berkelium.
The choice of name for both elements followed the same pattern: While Berkelium was named in honor of the University of Berkeley, the name Californium was chosen for the element 98 in honor of the university and the state of California: “ It is suggested that element 98 be given the name californium (symbol Cf) after the university and state where the work was done. This name, chosen for the reason given, does not reflect the observed chemical homology of element 98 to dysprosium (No. 66) as the names americium (No. 95), curium (No. 96), and berkelium (No. 97) signify that these elements are the chemical homologs of europium (No. 63), gadolinium (No. 64), and terbium (No. 65), respectively. ”
The sample was first prepared by applying curium nitrate solution (with the isotope 242 cm) to a platinum foil of about 0.5 cm 2 ; the solution was evaporated and the residue then calcined to oxide (CmO 2 ).
This sample was then bombarded in the 60-inch cyclotron with accelerated α-particles with an energy of 35 MeV for about 2-3 hours. In an (α, n) nuclear reaction, 245 Cf and free neutrons are produced :
After the bombardment in the cyclotron, the coating was dissolved using nitric acid, then again precipitated as the hydroxide using a concentrated aqueous ammonia solution ; the residue was dissolved in perchloric acid.
The further separation took place in the presence of a citric acid / ammonium citrate buffer in a weakly acidic medium ( pH ≈ 3.5) with ion exchangers at an elevated temperature.
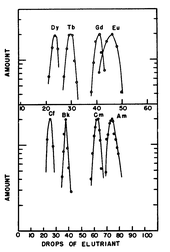
The chromatographic separation could only succeed on the basis of previous comparisons with the chemical behavior of the corresponding lanthanoids . In the event of a separation, the dysprosium emerges from a column before terbium , gadolinium and europium . If the chemical behavior of Californium is similar to that of an Eka-Dysprosium , the element in question 98 should therefore appear first in this analogous position, correspondingly before Berkelium, Curium and Americium.
The experiments also showed that only the +3 oxidation state was to be expected. Corresponding oxidation tests with ammonium peroxodisulfate or sodium bismuthate showed that either higher oxidation levels are not stable in aqueous solutions or that the oxidation itself is too slow.
The unequivocal identification succeeded when the previously calculated characteristic energy (7.1 MeV) of the α-particle emitted during the decay could be measured experimentally. The half-life of this α-decay was determined to be 45 minutes for the first time.
In the first publication it was assumed that the Californium isotope with the mass number 244 had been generated according to the following equation:
In 1956, this hypothesis was corrected: The 45-minute α-emitter, which was initially assigned to the isotope 244 Cf, was newly set to the mass number 245, which was determined, among other things, by long-term bombardment and decay studies. 245 Cf decays both through the emission of α-particles (7.11 ± 0.02 MeV) (≈ 30%) and through electron capture (≈ 70%). The new isotope 244 Cf was also determined and found that its decay takes place through the emission of an α-particle (7.17 ± 0.02 MeV with a half-life of 25 ± 3 minutes). The mass assignment of this isotope resulted from finding the Curium isotope 240 cm. The 244 Cf arises from the (α, 4n) reaction from 244 Cm:
In 1958, Burris B. Cunningham and Stanley G. Thompson isolated for the first time weighable amounts of a mixture of the isotopes 249 Cf, 250 Cf, 251 Cf, 252 Cf, which were produced by long-term neutron irradiation of 239 Pu in the test reactor at the National Reactor Testing Station in Idaho were. In 1960 BB Cunningham and James C. Wallmann isolated the first compound of the element, about 0.3 µg CfOCl, and then the oxide Cf 2 O 3 and the trichloride CfCl 3 .
Isotopes
There are 20 consistently radioactive isotopes and one core isomer of californium (mass numbers from 237 to 256). The longest lived are 251 Cf ( half-life 900 years), 249 Cf (351 years), 250 Cf (13 years), 252 Cf (2.645 years), and 248 Cf (334 days). The half-lives of the remaining isotopes range from milliseconds to hours or days.
If one takes out, for example, the decay of the longest-lived isotope 251 Cf, the long-lived 247 Cm is initially created through α-decay , which in turn changes to 243 Pu through renewed α-decay . The further decay then leads via 243 Am, 239 Np, 239 Pu to 235 U, the beginning of the uranium-actinium series (4 n + 3).
- The times given are half-lives.
The isotope 252 Cf decays with a half-life of 2.645 years to 96.908% by α-decay, but also to 3.092% by spontaneous fission . During spontaneous fission, an average of 3.77 neutrons are emitted per decaying nucleus. It is therefore used as a neutron source.
The isotope 254 Cf decays almost exclusively through spontaneous cleavage with a half-life of 60.5 days.
Occurrence
Californium isotopes do not occur naturally on earth because their half-life is too short compared to the age of the earth.
In the remains of the first American hydrogen bomb on November 1, 1952 on the Eniwetok Atoll - in addition to the first discovery of Einsteinium and Fermium and the detection of Plutonium and Americium - isotopes of Curium, Berkelium and Californium were found: especially the isotopes 245 cm and 246 cm, in smaller amounts 247 cm and 248 cm, in lanes 249 cm; furthermore 249 Bk, 249 Cf, 252 Cf, 253 Cf and 254 Cf. The amount of 249 Cf increased due to the β-decay of 249 Bk (half-life 330 days). In contrast, no 250 Cf was found. This can be attributed to the fact that the half-life of 250 Cm with around 8300 years is too long for a detectable amount of 250 Cf to be formed by β-decay (over 250 Bk) . In addition, 250 Cm only decays with a probability of around 6% in the β-decay to 250 Bk. For reasons of military secrecy, the results were only published in 1956.
It was assumed in the 1950s that Californium isotopes arise in the r process in supernovae . The isotope 254 Cf, which was previously found in the remains of the first American hydrogen bomb, was of particular interest . With the half-life measured at the time for spontaneous fission of 56.2 ± 0.7 days (currently: 60.5 days), it was assumed that there was a correspondence with the light curve of type I supernovae of 55 ± 1 days. However, the connection is still questionable.
The long-lived α-emitting isotopes 249 Cf and 251 Cf. Because of their long half-life, they are part of the transurane waste and are particularly problematic when it comes to final disposal.
Extraction and presentation
Californium is produced by bombarding lighter actinides with neutrons in a nuclear reactor . The main source is the 85 MW High-Flux-Isotope Reactor at Oak Ridge National Laboratory in Tennessee, USA, which is set up for the production of transcurium elements (Z> 96).
Extraction of californium isotopes
Californium is formed in nuclear reactors from uranium 238 U or plutonium isotopes through numerous successive neutron captures and β-decays - to the exclusion of fission or α-decays - which lead via Berkelium to the californium isotopes, first the isotopes with the mass numbers 249, 250, 251 and 252.
An important step here is the (n, γ) - or neutron capture reaction, in which the generated excited daughter nuclide changes to the ground state by emitting a γ quantum . The free neutrons required for this are created by fission of other nuclei in the reactor. In this nuclear chemical process, the plutonium isotope 239 Pu is initially formed by an (n, γ) reaction followed by two β - decays . In breeder reactors , this process is used to incubate new fissile material.
- The times given are half-lives .
For this purpose, the latter is irradiated with a neutron source that has a high neutron flux. The neutron fluxes possible here are many times higher than in a nuclear reactor. From 239 Pu is formed by four successive (n, γ) reactions 243 Pu, which decays to the americium isotope 243 Am by β-decay with a half-life of 4.96 hours . The 244 Am formed by a further (n, γ) reaction decays in turn to 244 Cm by β decay with a half-life of 10.1 hours . From 244 cm, further (n, γ) reactions in the reactor produce the next heavier isotopes in decreasing amounts.
However, the formation of 250 Cm in this way is very unlikely, since 249 Cm has only a short half-life and so further neutron captures are unlikely in the short time.
249 Cf is the first isotope of Californium that can be formed in this way. It is formed by two β-decays from 249 cm - the first curium isotope to undergo a β-decay (half-life 64.15 min).
The resulting here 249 Bk also forms by neutron capture the 250 Bk, which with a half-life of 3.212 hours through β-decay to Californiumisotop 250 decays Cf.
The isotopes 251 Cf, 252 Cf and 253 Cf are built up by further neutron capture . After one year of irradiation, the following isotope distribution is established: 249 Cf (2%), 250 Cf (15%), 251 Cf (4%) and 252 Cf (79%). The isotope 253 Cf decays to 253 Es with a half-life of 17.81 days .
Today californium is only available in very small quantities worldwide (mostly as oxide Cf 2 O 3 ), which is why it has a very high price. This is about 160 US dollars per microgram for 249 cf or 50 US dollars for 252 cf.
Californium ( 244 Cf and 246 Cf) was first extracted from uranium in 1951 by bombarding it with carbon:
The lighter isotopes of californium ( 240 Cf and 241 Cf) were created by bombarding 235 U, 234 U and 233 U with carbon in 1970.
Representation of elemental Californiums
Californium is obtained by reducing californium (III) oxide with lanthanum or thorium or of californium (III) fluoride with lithium or potassium .
In 1974 it was reported that californium was obtained for the first time in metallic form (a few micrograms) by reducing californium (III) oxide (Cf 2 O 3 ) with lanthanum and that the metal was applied in the form of thin films to supports for electron microscopy . Based on the measurements, an fcc structure ( a = 574.3 ± 0.6 pm ) and a hexagonal structure ( a = 398.8 ± 0.4 pm and c = 688.7 ± 0.8 pm) were initially described. The melting point was measured for the first time at 900 ± 30 ° C. However, these results were called into question in 1975. The two phases of californium were instead described as compounds of this metal: the hexagonal phase as Cf 2 O 2 S, the fcc phase as CfS. A trivalent californium with an atomic radius of 183–185 pm is described in both compounds. However, Noé and Peterson summarized the previous results in September 1975 and also presented their own extensive results that show the clear representation of metallic Californium and its properties.
properties
In the periodic table , californium with atomic number 98 is in the series of actinides, its predecessor is Berkelium , the following element is Einsteinium . Its analogue in the lanthanide series is dysprosium .
Physical Properties
Californium is a radioactive metal with a melting point of approx. 900 ° C and a density of 15.1 g / cm 3 . It occurs in three modifications : α-, β- and γ-Cf.
The α-Cf (<600 ° C) occurring under standard conditions crystallizes in the hexagonal crystal system in the space group P 6 3 / mmc (space group no.194) with the lattice parameters a = 338 pm and c = 1102.5 pm as well as four formula units per Unit cell . The crystal structure consists of a double hexagonal close packing of spheres (i.e. hcp) with the layer sequence ABAC and is therefore isotypic to the structure of α-La .
Under high pressure, α-Cf gradually changes to β-Cf. The β-modification (600–725 ° C) crystallizes in the cubic crystal system in the space group Fm 3 m (No. 225) with the lattice parameter a = 494 pm, which corresponds to a face-centered cubic lattice (fcc) or a cubic closest packing of spheres with the stacking sequence ABC corresponds to. Above 725 ° C the β-modification changes into the γ-modification. The γ-modification also crystallizes in the cubic crystal system, but with a larger lattice parameter of a = 575 pm.
The enthalpy of solution of californium metal in hydrochloric acid under standard conditions is −576.1 ± 3.1 kJ mol −1 . Based on this value, the standard enthalpy of formation (Δ f H 0 ) was calculated for the first time from Cf 3+ (aq) to −577 ± 5 kJ mol −1 and the standard potential Cf 3+ / Cf 0 to −1.92 ± 0, 03 V.
Chemical properties
Like all actinides, the shiny silver heavy metal is very reactive. It is attacked by water vapor, oxygen and acids; it is stable to alkalis.
The most stable oxidation level is the level +3 to be expected for the higher actinides. It forms two series of salts: Cf 3+ and CfO + compounds .
The two-valued and four-valued levels are also known. Cf (II) compounds are strong reducing agents . In water they release hydrogen with oxidation to Cf 3+ . Californium (IV) compounds are strong oxidizing agents. They are more unstable than those of curium and berkelium. So far, only two compounds of californium in the +4 oxidation state are known in solid form: californium (IV) oxide (CfO 2 ) and californium (IV) fluoride (CfF 4 ). Californium (IV) fluoride releases elemental fluorine when heated .
Aqueous solutions with Cf 3+ ions are green in color, with Cf 4+ ions they are brown.
In contrast to Cf 3+ ions, Cf 4+ ions are not stable in aqueous solution and can only be present in a complex-stabilized manner . The trivalent californium ( 249 Cf) was oxidized in potassium carbonate solution (K 2 CO 3 ) on a platinum anode. During the electrolysis, an increase in broadband absorption in the range of λ <500 nm could be observed (yellow coloration of the solution); the absorption band of Californium (III) decreased accordingly. Complete oxidation could not be achieved.
Cleavage
In general, all Californium isotopes with mass numbers between 249 and 254 are able to maintain a chain reaction with fission neutrons. For the other isotopes, the half-life is so short that not enough data on the behavior towards neutrons has been measured and published publicly. It is therefore not possible to calculate (as of 1/2009) whether a chain reaction with fast neutrons is possible, even if this is very likely. With thermal neutrons, a chain reaction succeeds in the isotopes 249, 251, 252, 253 and possibly 254. With the latter, the uncertainties of the current data are too great for a precise assessment (1/2009).
The isotope 251 Cf has a very small critical mass of only 5.46 kg for a pure sphere, which can be reduced to 2.45 kg with a reflector. This sparked speculation that it would be possible to build enormously small atom bombs . In addition to the very low availability and the associated high price, this is made more difficult by the short half-life of 251 Cf and the resulting high heat emission. The critical mass of 254 Cf, at around 4.3 kg, is still below that of 251 Cf, but the production of this isotope is significantly more complex and the half-life of 60.5 days is too short for use in nuclear weapons.
Furthermore, the isotopes 249 Cf, 251 Cf and 252 Cf would also be suitable for operating a nuclear reactor. In an aqueous solution with a reflector, the critical mass drops from 249 Cf to around 51 g, and that of 251 Cf even to around 20 g. All three isotopes could also be used in a fast reactor . This could also be achieved with 250 Cf (critical mass: 6.55 kg unreflected). However, this is countered by the low availability and the high price, which is why no reactors based on californium have been built so far. Accordingly, californium is not listed as a nuclear fuel in the German Atomic Energy Act .
safety instructions
Classifications according to the CLP regulation are not available because they only include the chemical hazard and play a completely subordinate role compared to the hazards based on radioactivity . The latter also only applies if the amount of substance involved is relevant.
use
Neutron source
The most interesting is the isotope 252 Cf. It disintegrates partly through spontaneous splitting ; 1 µg emits 2.314 million neutrons per second. It is therefore used exclusively for mobile, portable and at the same time powerful neutron sources ; for this purpose it is provided in the form of californium (III) oxide (Cf 2 O 3 ).
As a neutron source, it is used for:
- in medicine for cancer treatment
- in industry (material diagnostics, " On the Spot " - neutron activation analysis )
- for moisture measurement when drilling for crude oil (differentiation between water and oil-bearing layers)
- for finding explosives
- as a starting source in nuclear reactors
Manufacture of other items
For example, by bombarding 249 Cf with carbon, nobelium can be produced:
In October 2006 it was announced that the bombardment of 249 Cf with 48 Ca produced the heaviest element to date, Oganesson (element 118), after a previously announced discovery had been withdrawn.
links
Oxides
Californium exist oxides the oxidation states +3 (CF 2 O 3 ) and +4 (CFO 2 ).
Californium (IV) oxide (CfO 2 ) is produced by oxidation with molecular oxygen at high pressure and by atomic oxygen. It arises implicitly in nuclear reactors when irradiating uranium dioxide (UO 2 ) or plutonium dioxide (PuO 2 ) with neutrons. It is a black-brown solid and crystallizes - like the other actinide (IV) oxides - in the cubic crystal system in the fluorite structure . The lattice parameter is 531.0 ± 0.2 pm .
Californium (III) oxide (Cf 2 O 3 ) is a yellow-green solid with a melting point of 1750 ° C. There are two modifications; the transition temperature between the body-centered cubic and the monoclinic Cf 2 O 3 is around 1400 ° C. It is mainly used in the manufacture of 252 Cf neutron sources. For this purpose, 252 Cf (III) is first precipitated as californium oxalate (Cf 2 (C 2 O 4 ) 3 ), dried and then calcined to give a trivalent oxide.
Transitional compositions of oxides of the form CfO x (2.00> x> 1.50) have a rhombohedral structure.
Oxyhalides
Californium (III) oxifluoride (CfOF) was prepared by hydrolysis of californium (III) fluoride (CfF 3 ) at high temperatures. Like californium (IV) oxide (CfO 2 ), it crystallizes in the cubic crystal system in the fluorite structure, with the oxygen and fluorine atoms being found in random distribution on the anion positions. The lattice parameter is 556.1 ± 0.4 pm.
Californium (III) oxychloride (CfOCl) was prepared by hydrolysis of the hydrate of californium (III) chloride (CfCl 3 ) at 280-320 ° C. It has a tetragonal structure of the PbFCl type.
Halides
Halides are known for their oxidation states +2, +3 and +4. The most stable level +3 is known for all compounds from fluorine to iodine and is also stable in aqueous solution. The bivalent and tetravalent stage can only be stabilized in the solid phase.
| Oxidation number | F. | Cl | Br | I. |
| +4 |
Californium (IV) fluoride CfF 4 light green |
|||
| +3 |
Californium (III) fluoride CfF 3 yellow-green |
Californium (III) chloride CfCl 3 green |
Californium (III) bromide CfBr 3 green |
Californium (III) iodide CfI 3 red orange |
| +2 |
Californium (II) chloride CfCl 2 off- white |
Californium (II) bromide CfBr 2 amber |
Californium (II) iodide CfI 2 violet |
Californium (III) fluoride (CfF 3 ) is a yellow-green solid and has two crystalline structures that are temperature-dependent. At low temperatures, the orthorhombic structure of the YF 3 type can be found. At higher temperatures it forms a trigonal system of the LaF 3 type .
Californium (IV) fluoride (CfF 4 ) is a light green solid and crystallizes according to the monoclinic UF 4 type . Californium (IV) fluoride releases elemental fluorine when heated.
Californium (III) chloride (CFCl 3 ) is a green solid and forms two crystalline modifications: the hexagonal shape of UCl 3 type , wherein the Cf-9-atom is coordinated, as well as the orthorhombic form of the pUBR 3 type with the coordination number 8.
Californium (III) bromide (CfBr 3 ) is a green solid. Only the monoclinic structure of the AlCl 3 type could be detected. With increasing temperatures it partially decomposes to Californium (II) bromide (CfBr 2 ):
Californium (II) iodide (CfI 2 ) and californium (III) iodide (CfI 3 ) could be produced in microgram quantities in a high vacuum . These compounds were characterized by both X-ray diffraction and visible spectroscopy .
- CfI 3 is a red-orange solid and shows a rhombohedral structure of the BiI 3 type . The triiodide sublimes at ≈ 800 ° C without melting and can be prepared from Cf (OH) 3 and hydrogen iodide HI at 500 ° C.
- CfI 2 is a deep purple solid and is prepared from CfI 3 by reduction with H 2 at 570 ° C:
- It has two crystalline modifications: a rhombohedral structure of the CdCl 2 type that is stable at room temperature and a hexagonal, metastable structure of the CdI 2 type .
Pentelide
The pentelids of californium of the type CfX have been shown for the elements nitrogen , arsenic and antimony . They crystallize in the NaCl lattice with lattice constants 580.9 pm for CfAs and 616.6 pm for CfSb.
Organometallic compounds
Tri cyclopentadienyl complexes of the elements Berkelium (Cp 3 Bk) and Californium (Cp 3 Cf) are available from the trivalent stage. However, the high level of radioactivity causes rapid destruction of the connections.
literature
- Richard G. Haire: Californium , in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Eds.): The Chemistry of the Actinide and Transactinide Elements , Springer, Dordrecht 2006; ISBN 1-4020-3555-1 , pp. 1499-1576 ( doi : 10.1007 / 1-4020-3598-5_11 ).
- Gmelin's Handbook of Inorganic Chemistry , System No. 71, Transurane: Part A 1 I, pp. 40-43; Part A 1 II, pp. 19-20; Part A 2, pp. 222-233; Part B1, pp. 76-82.
- GT Seaborg (Ed.): Proceedings of the 'Symposium Commemorating the 25th Anniversary of the Discovery of Elements 97 and 98' , January 20, 1975; Report LBL-4366, July 1976.
- Harry H. Binder: Lexicon of chemical elements , S. Hirzel, Stuttgart 1999, ISBN 3-7776-0736-3 , pp. 139-142.
- Shot in the body . In: Der Spiegel . No. 17 , 1950, pp. 41 ( online ).
Web links
- Entry to Californium. In: Römpp Online . Georg Thieme Verlag, accessed on January 3, 2015.
- Jyllian Kemsley: Californium , Chemical & Engineering News, 2003.
- Pictures of CFBr 3 , CfOCl and Cf-metal (Engl.)
Individual evidence
- ↑ The values of the atomic and physical properties (Infobox) are, unless otherwise stated, taken from: Richard G. Haire: Californium , in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Ed.): The Chemistry of the Actinide and Transactinide Elements , Springer, Dordrecht 2006; ISBN 1-4020-3555-1 , pp. 1499-1576 ( doi : 10.1007 / 1-4020-3598-5_11 ).
- ↑ a b c d Binder, pp. 139–142.
- ↑ a b c d e Entry on californium in Kramida, A., Ralchenko, Yu., Reader, J. and NIST ASD Team (2019): NIST Atomic Spectra Database (ver. 5.7.1) . Ed .: NIST , Gaithersburg, MD. doi : 10.18434 / T4W30F ( https://physics.nist.gov/asd ). Retrieved June 13, 2020.
- ↑ a b c d e entry on californium at WebElements, https://www.webelements.com , accessed on June 13, 2020.
- ↑ a b c d e f g h i j k G. Audi, O. Bersillon, J. Blachot, AH Wapstra: The NUBASE evaluation of nuclear and decay properties . (pdf) In: Nuclear Physics A . 2003, No. 729, April, pp. 3-128. doi : 10.1016 / j.nuclphysa.2003.11.001 .
- ↑ The hazards emanating from radioactivity do not belong to the properties to be classified according to the GHS labeling. With regard to other hazards, this element has either not yet been classified or a reliable and citable source has not yet been found.
- ↑ SG Thompson, K. Street, Jr., A. Ghiorso, GT Seaborg: Element 98 , in: Physical Review , 1950 , 78 (3), pp. 298-299 ( doi: 10.1103 / PhysRev.78.298.2 ; Maschinoscript (February 27, 1950) ).
- ^ A b c S. G. Thompson, K. Street, Jr., A. Ghiorso, GT Seaborg: The New Element Californium (Atomic Number 98) , in: Physical Review , 1950 , 80 (5), pp. 790-796 ( doi : 10.1103 / PhysRev.80.790 ; abstract ; machine script (June 19, 1950) ) (PDF; 1.4 MB).
- ↑ K. Street, Jr., SG Thompson, GT Seaborg: Chemical Properties of Californium , in: J. Am. Chem. Soc. , 1950 , 72 (10), pp. 4832-4835 ( doi: 10.1021 / ja01166a528 ; printed by the United States Atomic Energy Commission (June 12, 1950) ).
- ↑ Alfred Chetham-Strode, Jr., Gregory R. Choppin, Bernard G. Harvey: Mass Assignment of the 44-Minute Californium-245 and the New Isotope Californium-244 , in: Physical Review , 1956 , 102 (3), p 747-748 ( doi: 10.1103 / PhysRev.102.747 ).
- ^ SG Thompson, BB Cunningham: First Macroscopic Observations of the Chemical Properties of Berkelium and Californium , supplement to Paper P / 825 presented at the Second Intl. Conf., Peaceful Uses Atomic Energy, Geneva, 1958.
- ↑ Darleane C. Hoffman, Albert Ghiorso, Glenn Theodore Seaborg: The Transuranium People: The Inside Story , Imperial College Press, 2000, ISBN 978-1-86094-087-3 , pp. 141-142 ( limited preview in Google Book search).
- ↑ Donald A. Hicks, John Ise, Jr., Robert V. Pyle: Multiplicity of Neutrons from the Spontaneous Fission of Californium-252 , in: Physical Review , 1955 , 97 (2), pp. 564-565 ( doi: 10.1103 /PhysRev.97.564 ).
- ↑ Donald A. Hicks, John Ise, Jr., Robert V. Pyle: Spontaneous-Fission Neutrons of Californium-252 and Curium-244 , in: Physical Review , 1955 , 98 (5), pp. 1521-1523 ( doi: 10.1103 / PhysRev.98.1521 ).
- ↑ Elis Hjalmar, Hilding Slätis, Stanley G. Thompson: Energy Spectrum of Neutrons from Spontaneous Fission of Californium-252 , in: Physical Review , 1955 , 100 (5), pp. 1542-1543 ( doi: 10.1103 / PhysRev.100.1542 ) .
- ↑ EJ Axton, BCNM Report GE / PH / 01/86 (June 1986).
- ↑ VV Golushko, KD Zhuravlev, Yu. S. Zamyatnin, NI Kroshkin, VN Nefedov: The average number of neutrons emitted in the spontaneous fission of Cm 244 , Cm 246 , and Cm 248 , in: Atomnaya Énergiya , 1973 , 34 (2), pp. 135-136 ( doi : 10.1007 / BF01163947 ).
- ^ PR Fields, MH Studier, H. Diamond, JF Mech, MG Inghram, GL Pyle, CM Stevens, S. Fried, WM Manning ( Argonne National Laboratory, Lemont, Illinois ); A. Ghiorso, SG Thompson, GH Higgins, GT Seaborg ( University of California, Berkeley, California ): Transplutonium Elements in Thermonuclear Test Debris , in: Physical Review , 1956 , 102 (1), pp. 180-182 ( doi: 10.1103 /PhysRev.102.180 ).
- ↑ JR Huizenga, H. Diamond: Spontaneous-Fission Half-Lives of Cf 254 and Cm 250 , in: Physical Review , 1957 , 107 (4), pp. 1087-1090 ( doi: 10.1103 / PhysRev.107.1087 ).
- ^ GR Burbidge , F. Hoyle (Mount Wilson and Palomar Observatories, Carnegie Institution of Washington, California Institute of Technology, Pasadena, California); EM Burbidge , Robert F. Christy , WA Fowler (Kellogg Radiation Laboratory, California Institute of Technology, Pasadena, California): Californium-254 and Supernovae , in: Physical Review , 1956 , 103 (5), pp. 1145-1149 ( doi : 10.1103 / PhysRev.103.1145 ; PDF ).
- ^ W. Baade , GR Burbidge , F. Hoyle , EM Burbidge , Robert F. Christy , WA Fowler : Supernovae and Californium 254 , in: Publications of the Astronomical Society of the Pacific , 1956 , 68 , No. 403, p. 296 -300 ( doi: 10.1086 / 126941 ; bibcode : 1956PASP ... 68..296B ).
- ↑ St. Temesváry: The element Californium-254 and the light curves of the supernovae of type I. A contribution to the question of the synthesis of heavy elements in the cosmos , in: Die Naturwissenschaften , 1957 , 44 (11), pp. 321–323 ( doi: 10.1007 / BF00630928 ).
- ↑ Edward Anders : Californium-254, Iron-59, and Supernovae of Type I , in: The Astrophysical Journal , 1959 , 129 , pp. 327-346 ( doi: 10.1086 / 146624 ; bibcode : 1959ApJ ... 129..327A ).
- ^ High Flux Isotope Reactor , Oak Ridge National Laboratory; Retrieved September 23, 2010.
- ↑ SG Thompson, A. Ghiorso, BG Harvey, GR Choppin: Transcurium Isotopes Produced in the Neutron Irradiation of Plutonium , in: Physical Review , 1954 , 93 (4), pp. 908-908 ( doi: 10.1103 / PhysRev.93.908 ) .
- ↑ H. Diamond, LB Magnusson, JF Mech, CM Stevens, AM Friedman, MH Studier, PR Fields, JR Huizenga: Identification of Californium Isotopes 249, 250, 251, and 252 from Pile-Irradiated Plutonium , in: Physical Review , 1954 , 94 (4), pp. 1083-1084 ( doi: 10.1103 / PhysRev.94.1083 ).
- ↑ LB Magnusson, MH Studier, PR Fields, CM Stevens, JF Mech, AM Friedman, H. Diamond, JR Huizenga: Berkelium and Californium Isotopes Produced in Neutron Irradiation of Plutonium , in: Physical Review , 1954 , 96 (6), p . 1576-1582 ( doi: 10.1103 / PhysRev.96.1576 ).
- ^ TA Eastwood, JP Butler, MJ Cabell, HG Jackson (Atomic Energy of Canada Limited, Chalk River, Ontario, Canada); RP Schuman, FM Rourke, TL Collins (Knolls Atomic Power Laboratory, Schenectady, New York): Isotopes of Berkelium and Californium Produced by Neutron Irradiation of Plutonium , in: Physical Review , 1957 , 107 (6), pp. 1635-1638 ( doi: 10.1103 / PhysRev.107.1635 ).
- ↑ Gmelin's Handbook of Inorganic Chemistry , System No. 71, Volume 7a, Transurane: Part A 1 II, pp. 19-20.
- ↑ Information on the element Californium at www.speclab.com (Engl.) ; Accessed September 22, 2008.
- ^ A. Ghiorso, SG Thompson, K. Street, Jr., GT Seaborg: Californium Isotopes from Bombardment of Uranium with Carbon Ions , in: Physical Review , 1951 , 81 (1), pp. 154–154 ( doi: 10.1103 / PhysRev . 81.154 ; Abstract ; Maschinoscript (September 6, 1950) ).
- ↑ RJ Silva, RL Hahn, KS Toth, ML Mallory, CE Bemis, Jr., PF Dittner, OL Keller: New Isotopes 241 Cf and 240 Cf , in: Physical Review C , 1970 , 2 (5), p. 1948– 1951 ( doi: 10.1103 / PhysRevC.2.1948 ).
- ↑ RG Haire, RD Baybarz: Crystal Structure and Melting Point of californium metal , in: J. Inorg. Nucl. Chem. , 1974 , 36 (6), pp. 1295-1302 ( doi: 10.1016 / 0022-1902 (74) 80067-9 ).
- ^ WH Zachariasen: On Californium Metal , in: J. Inorg. Nucl. Chem. , 1975 , 37 (6), pp. 1441-1442 ( doi: 10.1016 / 0022-1902 (75) 80787-1 ).
- ↑ a b c d M. Noé, JR Peterson: Preparation and Study of Elemental Californium-249 , in: Proceedings of the Fourth International Symposium on the Transplutonium Elements, Baden-Baden, 13. – 17. September 1975, North-Holland Publ. Co., Amsterdam 1975.
- ↑ VM Radchenko, AG Seleznev, RR Droznik, LS Lebedeva, MA Ryabinin, VD Shushakov, V. Ya. Vasil'ev & VT Ermishev, Soviet Radiochemistry (Engl. Transl.) , 1986 , 28 , p. 401; (Crystal structure at WebElements) .
- ↑ JR Peterson, U. Benedict, C. Dufour, I. Birkel, RG Haire: X-ray Diffraction Study of Californium Metal to 16 GPa , in: Journal of the Less Common Metals , 1983 , 93 (2), p. 353 -356 ( doi: 10.1016 / 0022-5088 (83) 90181-9 ).
- ↑ J. Fuger, RG Haire, JR Peterson: The Enthalpy of Solution of Californium Metal and the Standard Enthalpy of Formation of Cf 3+ (aq) , in: Journal of the Less Common Metals , 1984 , 98 (2), pp. 315-321 ( doi: 10.1016 / 0022-5088 (84) 90305-9 ).
- ^ DL Raschella, RG Haire, JR Peterson: First Determination of the Enthalpy of Solution of Californium Metal , in: Radiochim. Acta , 1982 , 30 , pp. 41-43.
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1956.
- ↑ BF Myasoedov, IA Lebedev, PL Khizhnyak, GA Timofeev, V. Ya. Frenkel: Electrochemical Oxidation of Americium and Californium in Carbonate Solutions , in: Journal of the Less Common Metals , 1986 , 122 , pp. 189-193 ( doi: 10.1016 / 0022-5088 (86) 90408-X ).
- ↑ V. Ya. Frenkel, Yu. M. Kulyako, VM Chistyakov, IA Lebedev, BF Myasoedov, GA Timofeev, EA Erin: Electrochemical Oxidation of Californium in Carbonate Solutions , in: Journal of Radioanalytical and Nuclear Chemistry , 1986 , 104 (4), pp. 191-199 ( doi : 10.1007 / BF02165322 ).
- ↑ MB Chadwick, P. Oblozinsky, M. Herman, NM Greene, RD McKnight, DL Smith, PG Young, RE MacFarlane, GM Hale, SC Frankle, AC Kahler, T. Kawano, RC Little, DG Madland, P. Moller, RD Mosteller, PR Page, P. Talou, H. Trellue, MC White, WB Wilson, R. Arcilla, CL Dunford, SF Mughabghab, B. Pritychenko, D. Rochman, AA Sonzogni, CR Lubitz, TH Trumbull, JP Weinman, DA Brown, DE Cullen, DP Heinrichs, DP McNabb, H. Derrien, ME Dunn, NM Larson, LC Leal, AD Carlson, RC Block, JB Briggs, ET Cheng, HC Huria, ML Zerkle, KS Kozier, A. Courcelle, V. Pronyaev, SC van der Marck: ENDF / B-VII.0: Next Generation Evaluated Nuclear Data Library for Nuclear Science and Technology , in: Nuclear Data Sheets , 2006 , 107 (12), pp. 2931-3060 ( doi: 10.1016 / j.nds.2006.11.001 ).
- ^ AJ Koning, et al .: The JEFF evaluated data project , Proceedings of the International Conference on Nuclear Data for Science and Technology, Nice 2007 ( doi: 10.1051 / ndata: 07476 ).
- ↑ a b c Institut de Radioprotection et de Sûreté Nucléaire : Evaluation of nuclear criticality safety data and limits for actinides in transport , p. 16 ( PDF ( Memento of November 18, 2014 in the Internet Archive )).
- ↑ Section 2, Paragraph 1 of the Atomic Energy Act, no supplementary ordinance relating to Californium available.
- ↑ K. Anderson, J. Pilcher, H. Wu, E. van der Bij, Z. Meggyesi, J. Adams: Neutron Irradiation Tests of an S-LINK-over-G-link System (November 21, 1999); PDF .
- ↑ RC Martin, JB Knauer, PA Balo: Production, Distribution, and Applications of Californium-252 Neutron Sources , 1999 ( PDF ).
- ↑ a b c R. C. Martin, JH Miller: Applications of Californium-252 Neutron Sources in Medicine, Research, and Industry , 2002 (?) ( PDF ( Memento of September 14, 2012 in the Internet Archive )).
- ↑ Patent US7118524 : Dosimetry for Californium-252 ( 252 Cf) Neutron-emitting Brachytherapy Sources and Encapsulation, Storage, and Clinical Delivery thereof. Filed April 22, 2003 , published October 10, 2006 , Applicant: New England Medical Center Hospitals, Inventor: Mark J. Rivard.
- ↑ H. Vogg, H. Braun: Neutron activation analysis with Californium-252 , lecture at the 4th seminar "Activation Analysis ", 11. – 12. October 1973, Berlin; PACT 45 report (1973).
- ↑ Bavarian State Ministry for the Environment, Health and Consumer Protection: Radioactivity and radiation measurement; 8th revised edition, April 2006 (PDF; 1.3 MB), p. 187.
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1954.
- ↑ Yu. Ts. Oganessian, VK Utyonkov, Yu. V. Lobanov, F. Sh. Abdullin, AN Polyakov, RN Sagaidak, IV Shirokovsky, Yu. S. Tsyganov, AA Voinov, GG Gulbekian, SL Bogomolov, BN Gikal, AN Mezentsev (Joint Institute for Nuclear Research, 141980 Dubna, Russian Federation); KJ Moody, JB Patin, DA Shaughnessy, MA Stoyer, NJ Stoyer, PA Wilk, JM Kenneally, JH Landrum, JF Wild, RW Lougheed (University of California, Lawrence Livermore National Laboratory , Livermore, California 94551, USA): Synthesis of the isotopes of elements 118 and 116 in the 249 Cf and 245 Cm + 48 Ca fusion reactions , in: Physical Review C , 2006 , 74 , pp. 044602-044610 ( doi: 10.1103 / PhysRevC.74.044602 ).
- ↑ a b c R. D. Baybarz, RG Haire, JA Fahey: On the Californium Oxide System , in: J. Inorg. Nucl. Chem. , 1972 , 34 (2), pp. 557-565 ( doi: 10.1016 / 0022-1902 (72) 80435-4 ).
- ^ A b J. C. Copeland, BB Cunningham: Crystallography of the Compounds of Californium. II. Crystal Structure and Lattice Parameters of Californium Oxychloride and Californium Sesquioxide , in: J. Inorg. Nucl. Chem. , 1969 , 31 (3), pp. 733-740 ( doi: 10.1016 / 0022-1902 (69) 80020-5 ).
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1972.
- ↑ Patent US3627691 : A method of preparing a Californium-252 neutron. Applied on January 8, 1970 , published December 14, 1971 , applicant: Atomic Energy Commission, inventor: Alexander R. Boulogne, Jean P. Faraci.
- ↑ JR Peterson, John H. Burns: Preparation and Crystal Structure of Californium Oxyfluoride, CfOF , in: J. Inorg. Nucl. Chem. , 1968 , 30 (11), pp. 2955-2958 ( doi: 10.1016 / 0022-1902 (68) 80155-1 ).
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 , p. 1969.
- ↑ JR Peterson, RL Fellows, JP Young, RG Haire: Stabilization of Californium (II) in the Solid State: Californium Dichloride, 249 CfCl 2 , in: Radiochem. Radioanal. Lett. , 1977 , 31 , pp. 277-282.
- ↑ JR Peterson, RD Baybarz: The Stabilization of Divalent Californium in the Solid State: Californium Dibromide , in: Inorg. Nucl. Chem. Lett. , 1972 , 8 (4), pp. 423-431 ( doi: 10.1016 / 0020-1650 (72) 80025-4 ).
- ↑ JN Stevenson, JR Peterson: The Trigonal and Orthorhombic Crystal Structures of CfF 3 and their Temperature Relationship , in: J. Inorg. Nucl. Chem. , 1973 , 35 (10), pp. 3481-3486 ( doi: 10.1016 / 0022-1902 (73) 80356-2 ).
- ↑ CT. P. Chang, RG Haire, SE Nave: Magnetic Susceptibility of Californium Fluorides , in: Physical Review B , 1990 , 41 (13), pp. 9045-9048 ( doi: 10.1103 / PhysRevB.41.9045 ).
- ↑ JH Burns, JR Peterson, RD Baybarz: Hexagonal and Orthorhombic Crystal Structures of Californium Trichloride , in: J. Inorg. Nucl. Chem. , 1973 , 35 (4), pp. 1171-1177 ( doi: 10.1016 / 0022-1902 (73) 80189-7 ).
- ↑ Californium bromide (CfBr 3 ): The crystal shows green color .
- ↑ JP Young, KL Vander Sluis, GK Werner, JR Peterson, M. Noé: High Temperature Spectroscopic and X-Ray Diffraction Studies of Californium Tribromide: Proof of Thermal Reduction to Californium (II) , in: J. Inorg. Nucl. Chem. , 1975 , 37 (12), pp. 2497-2501 ( doi: 10.1016 / 0022-1902 (75) 80878-5 ).
- ↑ a b c d J. F. Wild, EK Hulet, RW Lougheed, WN Hayes, JR Peterson, RL Fellows, JP Young: Studies of Californium (II) and (III) Iodides , in: J. Inorg. Nucl. Chem. , 1978 , 40 (5), pp. 811-817 ( doi: 10.1016 / 0022-1902 (78) 80157-2 ).
- ↑ a b c J. F. Wild, EK Hulet, RW Lougheed, WN Hayes: Preparation of Californium Diiodide , Symposium Commemorating the 25th Anniversary of the Discovery of Elements 97 and 98; January 20, 1975; Berkeley, USA.
- ^ A b J. E. Macintyre, FM Daniel, VM Stirling: Dictionary of inorganic compounds , Chapman and Hall, CRC Press, 1992, ISBN 978-0-412-30120-9 , p. 2826.
- ↑ D. Damien, RG Haire, JR Peterson: Californium-249 Monoarsenide and Monoantimonide , in: Inorg. Nucl. Chem. Lett. , 1980 , 16 (9-12), pp. 537-541 ( doi: 10.1016 / 0020-1650 (80) 80006-7 ).
- ↑ SE Nave, JR Moore, RG Haire, JR Peterson, DA Damien, Paul G. Huray: Magnetic Susceptibility of CfN, CfAs and CfSb , in: Journal of the Less Common Metals , 1986 , 121 , pp. 319-324 ( doi : 10.1016 / 0022-5088 (86) 90548-5 ).
- ↑ Christoph Elschenbroich : Organometallchemie , 6th edition, Wiesbaden 2008, ISBN 978-3-8351-0167-8 , pp. 583-584.
- ↑ Peter G. Laubereau, John H. Burns: Microchemical Preparation of Tricyclopentadienyl Compounds of Berkelium, Californium, and some Lanthanide Elements , in: Inorg. Chem. , 1970 , 9 (5), pp. 1091-1095 ( doi: 10.1021 / ic50087a018 ).





![{\ mathrm {^ {{251}} _ {{\ 98}} Cf \ {\ xrightarrow [{900 \ a}] {\ alpha}} \ _ {{\ 96}} ^ {{247}} cm \ {\ xrightarrow [{15 {,} 6 \ cdot 10 ^ {6} \ a}] {\ alpha}} \ _ {{\ 94}} ^ {{243}} Pu \ {\ xrightarrow [{4 {, } 956 \ h}] {\ beta ^ {-}}} \ _ {{\ 95}} ^ {{243}} On}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/2c4797edd762eb5b3cfa752d84a6308ae86241aa)
![{\ mathrm {^ {{238}} _ {{\ 92}} U \ {\ xrightarrow {(n, \ gamma)}} \ _ {{\ 92}} ^ {{239}} U \ {\ xrightarrow [{23.5 \ min}] {\ beta ^ {-}}} \ _ {{\ 93}} ^ {{239}} Np \ {\ xrightarrow [{2.3565 \ d}] {\ beta ^ {-}}} \ _ {{\ 94}} ^ {{239}} Pu}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/00b6f3352024666bbfe7ef4deba05e694195bfdf)
![{\ mathrm {^ {{239}} _ {{\ 94}} Pu \ {\ xrightarrow {4 (n, \ gamma)}} \ _ {{\ 94}} ^ {{243}} Pu \ {\ xrightarrow [{4,956 \ h}] {\ beta ^ {-}}} \ _ {{\ 95}} ^ {{243}} on \ {\ xrightarrow {(n, \ gamma)}} \ _ {{\ 95}} ^ {{244}} On \ {\ xrightarrow [{10.1 \ h}] {\ beta ^ {-}}} \ _ {{\ 96}} ^ {{244}} cm}} \ quad; \ quad {\ mathrm {^ {{244}} _ {{\ 96}} cm \ {\ xrightarrow {5 (n, \ gamma)}} \ _ {{\ 96}} ^ {{249}} Cm}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/20fba3c3fea74c7ee348a8b3c4a336a79b1537cf)
![{\ mathrm {^ {{249}} _ {{\ 96}} cm \ {\ xrightarrow [{64.15 \ min}] {\ beta ^ {-}}} \ _ {{\ 97}} ^ { {249}} Bk \ {\ xrightarrow [{330 \ d}] {\ beta ^ {-}}} \ _ {{\ 98}} ^ {{249}} Cf}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4a442eb6b9df6de89b3d39182cf49f3c45daaadc)
![{\ mathrm {^ {{249}} _ {{\ 97}} Bk \ {\ xrightarrow {(n, \ gamma)}} \ _ {{\ 97}} ^ {{250}} Bk \ {\ xrightarrow [{3,212 \ h}] {\ beta ^ {-}}} \ _ {{\ 98}} ^ {{250}} Cf}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d1ab240176cca76b16e7835a565f5b491a42cbc9)
![{\ mathrm {^ {{238}} _ {{\ 92}} U \ {\ xrightarrow [{-6 \ n}] {{\ mathrm {+ _ {{\ 6}} ^ {{12}} C }}}} \ _ {{\ 98}} ^ {{244}} Cf \ quad; \ quad _ {{\ 92}} ^ {{238}} U \ {\ xrightarrow [{-4 \ n}] {{\ mathrm {+ _ {{\ 6}} ^ {{12}} C}}}} \ _ {{\ 98}} ^ {{246}} Cf}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7290b676ed4fdc337c5baba10aea1402467ea8e7)




![{\ mathrm {^ {{249}} _ {{\ 98}} Cf \ {\ xrightarrow [{-2 \ n, \ - \ alpha}] {{\ mathrm {+ _ {{\ 6}} ^ { {12}} C}}}} \ _ {{102}} ^ {{255}} No}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/34a48bfb7c667ddfc1058628107da13e4b6c7fdd)

![{\ displaystyle {\ ce {2 CfF4 -> [\ Delta T] 2 CfF3 + F2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7766bbef3bf5a15682ca66af38d14d9f8c9f0d16)
![{\ displaystyle {\ ce {2 CfBr3 -> [\ Delta T] 2 CfBr2 + Br2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/062d1518dc82fcbfc0ee0fe5d9e89f34653c012d)
