Tantalum
| properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| General | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name , symbol , atomic number | Tantalum, Ta, 73 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Element category | Transition metals | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group , period , block | 5 , 6 , d | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Gray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | 7440-25-7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EC number | 231-135-5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECHA InfoCard | 100.028.304 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mass fraction of the earth's envelope | 8 ppm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic mass | 180,94788 (2) and | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius (calculated) | 145 (200) pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 138 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [ Xe ] 4 f 14 5 d 3 6 s 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1. Ionization energy | 7th.549 571 (25) eV ≈ 728.42 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2. Ionization energy | 16.2 (5) eV ≈ 1 560 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3. Ionization energy | 23.1 (1.6 eV) ≈ 2 230 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4. Ionization energy | 35.0 (1.7 eV) ≈ 3 380 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5. Ionization energy | 48.272 (25) eV ≈ 4 657.5 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physically | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical state | firmly | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | body-centered cubic | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| density | 16.65 g / cm 3 (20 ° C ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 6.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| magnetism | paramagnetic ( Χ m = 1.8 · 10 −4 ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 3290 K (3017 ° C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| boiling point | 5693 K (5420 ° C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar volume | 10.85 · 10 −6 m 3 · mol −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of evaporation | 753 kJ / mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 36 kJ mol −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound | 3400 m s −1 at 293.15 K. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Specific heat capacity | 140 J kg −1 K −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electric conductivity | 7.61 · 10 6 A · V −1 · m −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 57 W m −1 K −1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemically | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal potential | −0.81 V (½ Ta 2 O 5 + 5 H + + 5 e - → Ta + 2½ H 2 O) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | 1.5 ( Pauling scale ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| For other isotopes see list of isotopes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NMR properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAK |
Switzerland: 5 mg m −3 (measured as inhalable dust ) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tantalum [ ˈtʰantʰal ] is a chemical element with the symbol Ta and the atomic number 73; in the periodic table it is in the fifth subgroup or vanadium group . It is a rarely occurring, ductile , graphite-gray, shiny transition metal . Tantalum is mainly used for capacitors with high capacitance and at the same time small size. Since the metal is non-toxic and inert with regard to body fluids , it is also used for implants , for example for bone nails .
history

The element was found in 1802 by Anders Gustav Ekeberg both in a tantalite ore from Kimito in Finland and in ytter earth from Ytterby in Sweden . He separated a very stable oxide (tantalum (V) oxide) that did not dissolve in any acid. It is named after Tantalus , a figure from Greek mythology . Ekeberg chose this name to allude to the inability not to be able to absorb any of it, even with large amounts of acid.
A little earlier, in 1801, Charles Hatchett found an element he did not recognize in an ore from Massachusetts , which he named columbium . The oxides obtained from the two ores were compared in 1809 by William Hyde Wollaston and assumed to be oxides of a single element. He explained the measured differences in the specific gravity of the oxides by the different oxidation states of the element. Only Heinrich Rose distrusted this result and examined the ore detail. In 1844 he succeeded in proving that there are two different elements in the ores of the columbite-tantalite mixed crystal series . He named the lighter element found in the mineral columbite after Niobe , the daughter of Tantalus, niobium .
After the discovery of the new element, various chemists tried to represent tantalum in elemental form. The first to produce elemental tantalum by reducing potassium heptafluorotantalate with potassium was Jöns Jakob Berzelius in 1824 . However, like the tantalum represented by Rose, its metal was only 50% tantalum. In 1902, Henri Moissan tried to produce tantalum in an electric furnace, but the carbon it contained made his product very hard and brittle.
Werner von Bolton was the first to produce pure, ductile tantalum in 1903. He achieved this by reducing the glowing oxides in a vacuum and by melting impure tantalum metal in a vacuum and using an electric flame arc.
The first application of the new element was as a filament in light bulbs . The reason for the change from the previously used osmium to tantalum was that it is easier to process and has a higher possible usable temperature of up to 2300 ° C. It was later replaced by tungsten , which has an even higher melting point and thus enables a light spectrum that is closer to that of sunlight and a higher light output .
In 1922 a new application was found for tantalum with its use in rectifiers and a year later in radio tubes .
Occurrence
Tantalum is a rare element on earth with a content of 2 ppm in the continental crust and 8 ppm in the earth's shell. The frequency is comparable to that of arsenic and germanium . Within the group, the frequency decreases by a power of ten. In the solar system , tantalum is the rarest stable element.
Tantalum does not occur naturally , but only in the form of its compounds in various minerals. Due to the similarity of the two elements, tantalum ores always contain niobium and vice versa ( socialization ). The most important tantalum minerals are representatives of the columbite-tantalite mixed crystal series with the general formula (Fe, Mn) (Nb, Ta) 2 O 6 , which depending on the dominance of niobium or tantalum as columbite (mixed crystal series columbite (Fe) - columbite - (Mn) ) or tantalite (mixed crystal series tantalite (Fe) - tantalite (Mn) ), as well as representatives of the tapiolite series. Tantalum-rich columbites are also known as tantalites . Examples of tantalum-rich minerals of this series are tapiolite (Fe) , (Fe 2+ , Mn 2+ ) (Ta, Nb) 2 O 6 , and tantalite (Mn), Mn 2+ Ta 2 O 6 . These ores are often also called coltan . Coltan is a modern, primarily used in politics acronym from Col umbit - Tan talit, which denotes niobium-tantalum ores belonging to the four mixed crystal series columbite (Fe) - columbite (Mn), tantalite (Mn) - tantalite - (Fe), columbite (Mn) - tantalite (Mn) and columbite (Fe) - tantalite (Fe) count. Rarer tantalum-rich minerals are the representatives of the microlith group within the pyrochlore upper group and thoreaulite .
A large part of the tantalum supply in the western world, however, comes from slag that is produced during the smelting of tin ores, especially cassiterite . In 2011, Rwanda and the Democratic Republic of the Congo accounted for an estimated 40–50% of global tantalum mining . In 2007, however, Australia with 850 tons and Brazil with 250 tons were the most important producing countries of tantalum ores. Two mines in Australia were significant: the Wodgina Mine in the northwest and the mine in Bridgetown-Greenbushes Shire in Western Australia. Ores rich in tantalum can also be found in Canada and various African countries such as Ethiopia , Mozambique and Rwanda . The occurrences in the east of the Democratic Republic of the Congo , which were fiercely contested in the 1996-2008 Congo War and which continued to play an important role in the subsequent armed conflicts, became known in the media .
Some tantalum ores such as As the tantalum-rich representatives of columbite Tantalit- Mischkistallreihe were 2,012 American US by the Securities and Exchange Commission SEC as a so-called conflict minerals classified, its use is required to report for companies to the SEC. The reason given for this are the very production locations in eastern Congo, which are controlled by rebels and are therefore suspected of helping to finance armed conflicts.
Extraction and presentation
Since tantalum and niobium are always present together in the ores used for tantalum extraction, they have to be separated in order to extract the pure metals. This is made difficult by the great similarity between the two elements.
The first method of separation was developed by Jean Charles Galissard de Marignac in 1866 . He used the different solubility of the two elements in dilute hydrofluoric acid . Tantalum forms the slightly soluble K 2 TaF 7 , niobium the readily soluble K 3 NbOF 5 · 2 H 2 O.
The process used technically today is based on extraction and uses the different solubility of complex fluorine salts in water and certain organic solvents. The ore mixture is first dissolved in concentrated hydrofluoric acid or mixtures of hydrofluoric and sulfuric acid. The complex fluorides [NbOF 5 ] 2− and [TaF 7 ] 2− are formed . After insoluble constituents have been filtered off, the separation can be carried out by liquid-liquid extraction with the aid of methyl isobutyl ketone . If methyl isobutyl ketone is added to the solution, the niobium and tantalum complexes pass into the organic phase, while other elements such as iron or manganese remain in the aqueous phase. When water is added to the separated organic phase, only the niobium complex dissolves in it, the tantalum remains in the methyl isobutyl ketone.
The tantalum can be precipitated with the help of potassium fluoride as poorly soluble potassium heptafluorotantalate K 2 [TaF 7 ]. The reduction to elemental tantalum is usually done by sodium .
- Reduction with sodium
A possible alternative to extraction is fractional distillation . The different boiling points of the two chlorides niobium (V) chloride and tantalum (V) chloride are used for this purpose. These can be extracted from the ores with chlorine and coke at high temperatures . After the separation, the tantalum chloride is also reduced to the metal with sodium.
In addition to columbite-tantalite ores, slag from tin smelting is an important source for tantalum extraction (contains a few percent tantalum).
recycling
Tantalum from capacitors in electronic assemblies is almost never recycled. In contrast, the internal waste generated in the manufacture of powders and capacitors can be recycled. During the manufacture of tantalum rolled products (20% of consumption), chips and faulty batches are generated, which can be effectively recycled by remelting. In addition, tantalum carbide alloy additives are recyclable.
Tantalum (V) oxide from glasses and such in the form of lithium tantalate single crystals in electronic components requires a complex chemical recycling process. This includes roasting , dissolving in hydrofluoric acid or sulfuric acid, solvent extraction with ketones and the precipitation of tantalum (V) oxide or the crystallization of potassium fluorotantalate . These are then melted in the electron beam furnace.
properties
Physical Properties
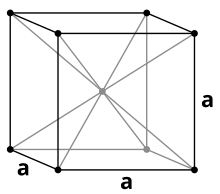
Tantalum is a distinctly purple-gray, steel-hard ( Vickers hardness : 60–120 HV), high-melting heavy metal that is similar to niobium in most of its properties . It crystallizes in a body-centered cubic crystal structure . In addition to the cubic α-structure, β-tantalum is also known, which crystallizes in a tetragonal crystal structure corresponding to β-uranium with the lattice parameters a = 1021 pm and c = 531 pm. This modification is metastable and can be obtained by electrolysis of a tantalum fluoride melt.
With a melting point of around 3000 ° C, tantalum has the highest melting point of all elements after tungsten , carbon and rhenium . If only a small amount of carbon or hydrogen is stored in the metal , the melting point increases significantly. A substoichiometric tantalum carbide has one of the highest melting points of all substances with a melting point of 3983 ° C.
Tantalum becomes a superconductor below a transition temperature of 4.3 Kelvin .
While pure tantalum is ductile and can be stretched considerably ( tensile strength : 240 MPa), even small amounts of carbon or hydrogen added to it change the mechanical strength significantly. The material becomes brittle and difficult to process. This fact is used to produce tantalum powder. In technology, it is loaded with hydrogen and thus embrittled, then comminuted accordingly and, at a higher temperature, freed from hydrogen again by heating .
Chemical properties
Tantalum is a base metal and reacts at high temperatures with most non-metals , such as oxygen , the halogens or carbon. At room temperature, however, the metal is protected by a thin layer of tantalum (V) oxide and thus passivated . A reaction only takes place from a temperature of around 300 ° C. As a powder, it is a flammable solid that can be easily ignited by brief exposure to an ignition source and then continues to burn after its removal. The more finely the substance is distributed, the greater the risk of ignition. The metal in compact form is not flammable.
Because of its passivation, tantalum is not soluble in most acids ; even aqua regia cannot dissolve the metal. Tantalum is only attacked by hydrofluoric acid , oleum (a mixture of sulfuric acid and sulfur trioxide ) and molten salt .
Isotopes
A total of 30 isotopes and 26 core isomers from 155 Ta to 185 Ta are known. Natural tantalum consists almost exclusively (99.988%) of the isotope 181 Ta. In addition, 0.012% of the core isomer 180m Ta occurs.
180m Ta is the only long-lived, naturally occurring nuclide that is not in its ground state , but in an excited state. No radioactive decay has been observed so far, the half-life of the isomer must be at least 2 · 10 16 years. The ground state 180 Ta, on the other hand, is unstable and decays with a half-life of only 8.125 hours. 180m Ta has a frequency of 2.49 · 10 −6 in the solar system (based on silicon = 1 · 10 6 ).
use
Most of the tantalum (worldwide annual production volume 1,400 t) is used for very small capacitors with high capacitance . In 2007, 60% of the tantalum was used in the manufacture of capacitors. These tantalum electrolytic capacitors are used everywhere in modern microelectronics , for example for cell phones and in automobile construction. The effect is based on the tantalum oxide layer on the surface of the wound tantalum foil, which is still stable and reliably insulating even in a very thin version. The thinner the layer between the electrodes, the higher the capacity with the same foil surface; In addition, tantalum oxide has an extremely high permittivity , which also increases the capacity.
Since tantalum is non-toxic and does not react with body tissue or fluids, elemental tantalum is used for medical implants and instruments. For example, bone nails , prostheses , clips and jaw screws are made from tantalum. As a coating on a porous carbon grid, it achieves particularly good osseointegration , which is why tantalum-coated implants are used primarily in reconstruction surgery for larger tumor or infection-related loss of bone substance, without having an antimicrobial effect. In addition, it is an X-ray contrast medium that is little used because of its high costs .
In the chemical industry, tantalum is used because of its durability. It serves as a lining material for reaction vessels and is used for heat exchangers and pumps . For these purposes, pure tantalum is usually not used, but alloys that contain 2.5–10% tungsten . These are more stable and resistant than pure tantalum. At the same time, the desired ductility is retained. Other uses are laboratory equipment , spinnerets and the cathodes of electron tubes . Here, tantalum benefits from the fact that it is able to absorb up to 740 parts by volume of gases at 800 ° C ( getter effect ), which ensures a high vacuum in the tubes.
Superalloys , which are used in the construction of turbines and aircraft engines, contain up to 9% tantalum. Adding 3–4% tantalum to a nickel superalloy increases the strength of the material at high temperatures.
safety instructions
Working with tantalum and its compounds normally does not cause any problems under laboratory conditions. Elementary tantalum and tantalum compounds are not toxic. However, there are vague indications that some tantalum compounds are carcinogenic. Tantalum powder and dust - like other finely divided metals - pose a high risk of fire and explosion .
links
Tantalum (V) oxide Ta 2 O 5 is a white powder that is used to manufacture highly refractive glasses and special crystal materials.
Tantalum carbide TaC, with its melting temperature of 3880 ° C and a hardness similar to that of quartz, serves as a protective layer on high-temperature alloys in engines and cutting tools.
literature
- AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 .
- Norman N. Greenwood, Alan Earnshaw: Chemistry of the Elements. 1st edition. Wiley-VCH, Weinheim 1988, ISBN 3-527-26169-9 .
- Klaus Andersson, Karlheinz Reichert, Rüdiger Wolf: Tantalum and Tantalum Compounds. In: Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH, 2000, doi : 10.1002 / 14356007.a26_071 .
- Harry H. Binder: Lexicon of the chemical elements - the periodic table in facts, figures and data. Hirzel, Stuttgart 1999, ISBN 3-7776-0736-3 .
Web links
Individual evidence
- ^ Harry H. Binder: Lexicon of the chemical elements. S. Hirzel Verlag, Stuttgart 1999, ISBN 3-7776-0736-3 .
- ↑ The values for the properties (info box) are taken from www.webelements.com (tantalum) unless otherwise stated .
- ↑ CIAAW, Standard Atomic Weights Revised 2013 .
- ↑ a b c d e Entry on tantalum in Kramida, A., Ralchenko, Yu., Reader, J. and NIST ASD Team (2019): NIST Atomic Spectra Database (ver. 5.7.1) . Ed .: NIST , Gaithersburg, MD. doi : 10.18434 / T4W30F ( https://physics.nist.gov/asd ). Retrieved June 13, 2020.
- ↑ a b c d e Entry on tantalum at WebElements, https://www.webelements.com , accessed on June 13, 2020.
- ^ NN Greenwood, A. Earnshaw: Chemistry of the elements. 1st edition. VCH, Weinheim 1988, ISBN 3-527-26169-9 , p. 1260.
- ↑ Robert C. Weast (Ed.): CRC Handbook of Chemistry and Physics . CRC (Chemical Rubber Publishing Company), Boca Raton 1990, ISBN 0-8493-0470-9 , pp. E-129 to E-145. Values there are based on g / mol and given in cgs units. The value specified here is the SI value calculated from it, without a unit of measure.
- ↑ a b Yiming Zhang, Julian RG Evans, Shoufeng Yang: Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. In: Journal of Chemical & Engineering Data . 56, 2011, pp. 328-337, doi: 10.1021 / je1011086 .
- ↑ a b c Entry on tantalum, powder in the GESTIS substance database of the IFA , accessed on April 30, 2017(JavaScript required) .
- ↑ Swiss Accident Insurance Fund (Suva): Limit values - current MAK and BAT values (search for 7440-25-7 or tantalum ), accessed on November 2, 2015.
- ↑ Anders Gustav Ekeberg: About a new metal, tantalum, which was discovered in some Swedish fossils at the same time as the ytter earth; together with some remarks about the properties of ytter earth, in comparison with beryl earth. In: Crell's Annals of Chemistry . 1, 1803, pp. 1–21 ( limited preview in Google Book search).
- ^ C. Hatchett: An Analysis of a Mineral Substance from North America, Containing a Metal Hitherto Unknown. In: Philosophical Transactions of the Royal Society of London . 92, 1802, pp. 49-66, doi: 10.1098 / rstl.1802.0005 .
- ^ WH Wollaston: On the Identity of Columbium and Tantalum. In: Philosophical Transactions of the Royal Society of London . 99, 1809, pp. 246-252, doi: 10.1098 / rstl.1809.0017 .
- ↑ Heinrich Rose: About the composition of tantalite and a new metal contained in the tantalite of Bavaria. In: Annals of Physics . 139, 10, 1844, pp. 317-341 ( digitized on Gallica ).
- ↑ JJ Berzelius: hydrofluoric acid tantalic acid and hydrofluoric acid tantalum salts, tantalum and various of its compounds. In: Annals of Physics and Chemistry . 4, 1825, pp. 6-22 ( digitized on Gallica ).
- ↑ Werner von Bolton: The tantalum, its representation and its properties. In: Z. f. Electrochem. 11, 1905, pp. 45-52 (Referat in: Angew. Chem. , 1905, 18, pp. 1451-1466, doi: 10.1002 / anie.19050183603 ).
- ^ Henri Moissan: Sur la preparation du tantale au four electrique et sur ses propriétés. In: Comptes Rendus . 134, 1902, pp. 211-215 ( digitized on Gallica ).
- ↑ Entry on tantalum. In: Römpp Online . Georg Thieme Verlag, accessed on April 19, 2014.
- ↑ a b Ziemann: The tantalum lamp . Lecture at the Association of German Chemists, Halle (Saale), March 19, 1905, In: Angew. Chem. 18, 1905, pp. 790-800, doi: 10.1002 / anie.19050182005 .
- ↑ SI Venetzkij: tales of metals. Leipzig 1988, ISBN 3-342-00324-3 .
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 85th edition. CRC Press, Boca Raton, Florida 2005. Section 14, Geophysics, Astronomy, and Acoustics; Abundance of Elements in the Earth's Crust and in the Sea.
- ↑ a b J. Laeter, N. Bukilic: Isotope abundance of 180 Ta m and p-process nucleosynthesis. In: Physical Review C . 72, 2005, S., doi: 10.1103 / PhysRevC.72.025801 .
- ↑ TS Ercit, MA Wise, R. Černý: compositional and structural systematics of the columbite group. In: American Mineralogist . 80, 1995, pp. 613–619 (PDF)
- ↑ ddc.arte.tv
- ↑ a b Tantalum Production 2007 (PDF; 86 kB) published by the United States Geological Survey .
- ^ War in Congo: On the dark side of the digital world. In: Frankfurter Allgemeine Zeitung . August 23, 2010.
- ↑ Benjamin Dürr: The stuff that cell phones are made of. In: stern.de. March 16, 2010.
- ↑ 1990-1993: US Geological Survey, 1994 Minerals Yearbook . (PDF; 174 kB). (MYB), "COLUMBIUM (NIOBIUM) AND TANTALUM" by Larry D. Cunningham, Table 10; 1994–1997: MYB 1998 (PDF; 79 kB) Table 10; 1998–2001: MYB 2002 (PDF; 322 kB) p. 21.13; 2002–2003: MYB 2004 (PDF; 599 kB) p. 20.13; 2004: MYB 2008 (PDF; 76 kB) p. 52.12; 2005–2009: MYB 2009 (PDF; 132 kB) p. 52.13. USGS did not report data for other countries (China, Kazakhstan, Russia, etc.) owing to data uncertainties.
- ^ SEC: Conflict Minerals - Final Rule. 2012, pp. 34–35, (PDF, 1.96 MB, engl.)
- ↑ SEC Adopts Rule for Disclosing Use of Conflict Minerals. Accessed September 3, 2012.
- ^ Jean Charles Galissard de Marignac: Recherches sur les combinaisons du niobium. In: Annales de chimie et de physique . 4, 1866, pp. 5-75 ( digitized on Gallica ).
- ↑ Joachim Eckert: Niobium and Niobium Compounds. In: Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH, 2000, doi : 10.1002 / 14356007.a17_251 .
- ↑ Entry on tantalum recycling. In: Römpp Online . Georg Thieme Verlag, accessed on April 19, 2014.
- ↑ K. Schubert: A model for the crystal structures of the chemical elements. In: Acta Crystallographica . B30, 1974, pp. 193-204.
- ↑ a b c d e f g Klaus Andersson, Karlheinz Reichert, Rüdiger Wolf: Tantalum and Tantalum Compounds. In: Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH, 2000.
- ↑ Alla Arakcheeva, Gervais Chapuis, Vladimir Grinevitch: The self-hosting structure of β-Ta. In: Acta Crystallographica Section B Structural Science. 58, 2002, pp. 1-7, doi: 10.1107 / S0108768101017918 .
- ↑ L. Malter, D. Langmuir: Resistance, Emissivities and Melting Point of Tantalum. In: Physical Review . 55, 1939, pp. 743-747, doi: 10.1103 / PhysRev.55.743 .
- ^ DJ Rowcliffe, WJ Warren: Structure and properties of tantalum carbide crystals. In: Journal of Materials Science . 5, 1970, pp. 345-350, doi: 10.1007 / BF02397788 .
- ↑ G. Audi, O. Bersillon, J. Blachot, AH Wapstra: The NUBASE evaluation of nuclear and decay properties. In: Nuclear Physics. Volume A 729, 2003, pp. 3-128. doi : 10.1016 / j.nuclphysa.2003.11.001 . ( PDF ; 1.0 MB).
- ↑ Mikael Hult, JS Elisabeth Wieslander, Gerd Marissens, Jo l Gasparro, Uwe Waetjen, Marcin Misiaszek: Search for the radioactivity of 180m Ta using an underground HPGe sandwich spectrometer. In: Applied Radiation and Isotopes . 67, 2009, pp. 918-921, doi: 10.1016 / j.apradiso.2009.01.057 .
- ^ PL Harrison, T. Harrison, I. Stockley, TJ Smith: Does Tantalum exhibit any intrinsic antimicrobial or antibiofilm properties? The Bone & Joint Journal 2017, Volume 99-B, Issue 9 from September 2017 pages 1153–1156, [DOI: 10.1302 / 0301-620X.99B9.BJJ-2016-1309.R1]
- ↑ E. Kammler, WT Ulmer: A new way in bronchography - On the representation of the Trascheobronchialbaumes in animals by inhalation of tantalum dust. In: Pneumology . Edition 144, Volume 4, Springer-Verlag 1971, pp. 344–351.



![\ mathrm {K_ {2} [TaF_ {7}] + 5 \ Na \ longrightarrow Ta + 5 \ NaF + 2 \ KF}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a8e5249d029b3b11174bfa47836de66d65159664)