sodium
| properties | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| General | ||||||||||||||||||||||||||||||||||||||||
| Name , symbol , atomic number | Sodium, Na, 11 | |||||||||||||||||||||||||||||||||||||||
| Element category | Alkali metals | |||||||||||||||||||||||||||||||||||||||
| Group , period , block | 1 , 3 , p | |||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white | |||||||||||||||||||||||||||||||||||||||
| CAS number | 7440-23-5 | |||||||||||||||||||||||||||||||||||||||
| EC number | 231-132-9 | |||||||||||||||||||||||||||||||||||||||
| ECHA InfoCard | 100.028.302 | |||||||||||||||||||||||||||||||||||||||
| Mass fraction of the earth's envelope | 2.64% | |||||||||||||||||||||||||||||||||||||||
| Atomic | ||||||||||||||||||||||||||||||||||||||||
| Atomic mass | 22.98976928 (2) et al | |||||||||||||||||||||||||||||||||||||||
| Atomic radius (calculated) | 180 (190) pm | |||||||||||||||||||||||||||||||||||||||
| Covalent radius | 154 pm | |||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 227 pm | |||||||||||||||||||||||||||||||||||||||
| Electron configuration | [ Ne ] 3 s 1 | |||||||||||||||||||||||||||||||||||||||
| 1. Ionization energy | 5.139 076 9 (3) eV ≈ 495.85 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| 2. Ionization energy | 47.28636 (25) eV ≈ 4 562.44 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| 3. Ionization energy | 71.6200 (12) eV ≈ 6 910.28 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| 4. Ionization energy | 98.936 (12) eV ≈ 9 545.87 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| 5. Ionization energy | 138.404 (12) eV ≈ 13 354 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| Physically | ||||||||||||||||||||||||||||||||||||||||
| Physical state | firmly | |||||||||||||||||||||||||||||||||||||||
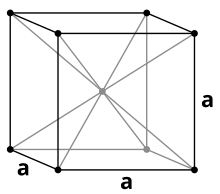
| Crystal structure | body-centered cubic | |||||||||||||||||||||||||||||||||||||||
| density | 0.968 g / cm 3 (20 ° C ) | |||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 0.5 | |||||||||||||||||||||||||||||||||||||||
| magnetism | paramagnetic ( Χ m = 8.5 10 −6 ) | |||||||||||||||||||||||||||||||||||||||
| Melting point | 370.87 K (97.72 ° C) | |||||||||||||||||||||||||||||||||||||||
| boiling point | 1163 K (890 ° C) | |||||||||||||||||||||||||||||||||||||||
| Molar volume | 23.78 10 −6 m 3 mol −1 | |||||||||||||||||||||||||||||||||||||||
| Heat of evaporation | 97.4 kJ / mol | |||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 2.6 kJ mol −1 | |||||||||||||||||||||||||||||||||||||||
| Vapor pressure | 1 Pa at 554 K | |||||||||||||||||||||||||||||||||||||||
| Speed of sound | 3200 m s −1 at 293.15 K. | |||||||||||||||||||||||||||||||||||||||
| Specific heat capacity | 1230 J kg −1 K −1 | |||||||||||||||||||||||||||||||||||||||
| Work function | 2.75 eV | |||||||||||||||||||||||||||||||||||||||
| Electric conductivity | 21 · 10 6 A · V −1 · m −1 | |||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 140 W m −1 K −1 | |||||||||||||||||||||||||||||||||||||||
| Chemically | ||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 1 | |||||||||||||||||||||||||||||||||||||||
| Normal potential | −2.713 V (Na + + e - → Na) | |||||||||||||||||||||||||||||||||||||||
| Electronegativity | 0.93 ( Pauling scale ) | |||||||||||||||||||||||||||||||||||||||
| Isotopes | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| For other isotopes see list of isotopes | ||||||||||||||||||||||||||||||||||||||||
| NMR properties | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . |
||||||||||||||||||||||||||||||||||||||||
Sodium is a frequently occurring chemical element with the symbol Na and the atomic number 11. In the periodic table of the elements it is in the 3rd period and as an alkali metal in the 1st IUPAC group or 1st main group . Sodium is a pure element whose only stable isotope 23 is Na.
Elemental sodium was first in 1807 by Humphry Davy by fused salt electrolysis of sodium hydroxide recovered and Sodium called. This term is used in the English and French-speaking areas, derivatives of it in the Romance and partly also in Slavic languages. The German name sodium is Arabic نطرون, DMG naṭrūn , soda, derived from the Egyptian netjerj . Sodium and its derivatives are used in Scandinavia, Dutch and some Slavic languages as well as the German language. In Japanese, sodium has the German-sounding name Japanese ナ ト リ ウ ム Natoriumu .
Under normal conditions , sodium is a waxy, silvery, highly reactive metal. Because of its strong reactivity , metallic (elemental) sodium is stored under inert conditions, mostly in paraffin oil or petroleum , with larger quantities in airtight steel drums.
Sodium is one of the ten most common elements in the earth's shell and occurs in numerous minerals in the earth's crust . In seawater is a significant amount of sodium in the form of sodium ion contained.
history
The Egyptians coined in ancient times for the derived soda lakes soda the term netjerj ( neter ). The Greeks adopted this word as Greek νίτρον nitron , the Romans as nitrium , the Arabs as natrun . In contrast to elementary metal, sodium compounds have been known for a very long time and have since been extracted from sea water or lakes, mined from underground deposits and traded. The most important sodium compound, table salt ( sodium chloride ), was obtained in mines or by drying sea water or salty spring water in salt pans . The trade in salt was the basis of their wealth for many cities and sometimes even shaped their names ( Salzgitter , Salzburg ). Place names such as Hallstatt , Hallein , Halle (Saale) , Bad Hall , Bad Reichenhall , Schwäbisch Hall , Schweizerhalle or Hall in Tirol allude to the Germanic name for Saline (Hall) . Other naturally occurring sodium compounds such as sodium carbonate ( soda or baking soda ) and sodium nitrate have also been extracted and traded since ancient times.
The production of elemental sodium only succeeded in 1807 Humphry Davy by electrolysis of molten sodium hydroxide ( caustic soda ) using voltaic columns as a power source. As he reported to the Royal Society in London on November 19, 1807 , he won two different metals: he called the sodium contained in soda sodium , which is the name still used in French and English-speaking countries; he called the other metal Potassium ( potassium ). In 1811 Berzelius proposed the current name sodium .
Occurrence
In the universe, sodium ranks 14th in frequency, comparable to calcium and nickel . The yellow sodium D line can be easily detected in the light emitted by many celestial bodies , including that of the sun .
Sodium is the sixth most common element on earth, accounting for 2.36% of the earth's crust . Due to its reactivity, it does not occur in elemental form, but always in compounds, the sodium salts . Sea water is a large reservoir of sodium . One liter of seawater contains an average of 11 grams of sodium ions.
Common sodium minerals are albite (called soda feldspar), NaAlSi 3 O 8 and oligoclase (Na, Ca) Al (Si, Al) 3 O 8 . In addition to these rock-forming minerals, which belong to the feldspars , sodium occurs in large salt deposits . Above all, there are large deposits of halite ( sodium chloride , colloquially often called rock salt), which were created by the drying out of parts of the sea. These represent the most important source for the extraction of sodium and its compounds. Well-known German salt production sites include Salzgitter , Bad Reichenhall , Stade and Bad Friedrichshall .
In addition to the common sodium chloride, other compounds occur in nature. Thus, sodium nitrate or sodium nitrate (also known as Chilean nitrate) NaNO 3 one of the few natural nitrate minerals . Because of its good solubility in water, it occurs only in particularly dry regions, such as the Atacama Desert in Chile . Before the Haber-Bosch process was invented , this was the most important raw material for many fertilizers and explosives .
Sodium carbonate Na 2 CO 3 is also found naturally in several minerals. The best-known mineral is soda Na 2 CO 3 · 10 H 2 O. It is mined in large quantities and mainly used in glass production.
There are also a number of other sodium minerals (see also: Category: Sodium Minerals ). A well-known one is cryolite ( ice stone , Na 3 [AlF 6 ]), which in its molten state serves as a solvent for aluminum oxide in aluminum production. Since the only known cryolite deposit is mined in Greenland , cryolite is artificially produced.
Extraction and presentation
Sodium is mainly obtained from sodium chloride, which is usually obtained by mining or by drying out saline solutions such as seawater. Only a small part of the sodium chloride is processed into elemental sodium, the greater part is used as table salt or for the production of other sodium compounds.
The large-scale production of sodium is carried out by fused- salt electrolysis of dry sodium chloride in a so-called Downs cell (patented in 1924 by James C. Downs ). For lowering the melting point a is eutectic salt mixture of 60% calcium chloride and 40% sodium chloride used which melts at 580 ° C. Also barium is possible as an addition. A voltage of about seven volts is applied. To produce one kilogram of sodium, around 10 kWh of electricity are used during electrolysis, and around 12 kWh for the entire production process.
- Formation of sodium on the cathode
- Formation of chlorine on the anode
- Overall response
The cylindrical electrolysis cell consists of a central graphite anode and a lateral iron cathode ring . Above the cell is a bell that collects and discharges the chlorine. The sodium collects above the cathodes and is removed from the cell through a cooled riser pipe. Also Resulting calcium crystallizes out there and fall back into the melt.
The electrolysis of sodium chloride replaced the Castner process . The sodium was obtained by fused-salt electrolysis of sodium hydroxide. This had the advantage of the lower melting point of sodium hydroxide (318 ° C), but more electrical energy is required. Since the introduction of chlor-alkali fused-salt electrolysis, the price of sodium has fallen dramatically. In terms of volume, sodium is thus the cheapest light metal of all. However, the price depends heavily on the electricity costs and the price of the chlorine that is also produced .
properties
Physical Properties
Sodium is a silvery white, soft light metal . In many properties it stands between lithium and potassium. The melting point of 97.82 ° C is between that of lithium (180.54 ° C) and that of potassium (63.6 ° C). This is similar with the boiling point and the specific heat capacity . With a density of 0.968 g · cm −3 , sodium is one of the specifically lightest elements. Of the elements that are solid at room temperature, only lithium and potassium have a lower density. With a Mohs hardness of 0.5, sodium is so soft that it can be cut with a knife.
Sodium crystallizes, like the other alkali metals, in the cubic crystal system in a body- centered lattice with the space group Im 3 m (space group no. 229) and two formula units per unit cell . Below 51 K it changes into a hexagonal closest packing of spheres with the lattice parameters a = 376 pm and c = 615 pm.
Sodium vapor consists of both individual metal atoms and dimers of the form Na 2 . At the boiling point, 16% of the atoms are in the form of dimers. The vapor is yellow and appears purple when viewed through.
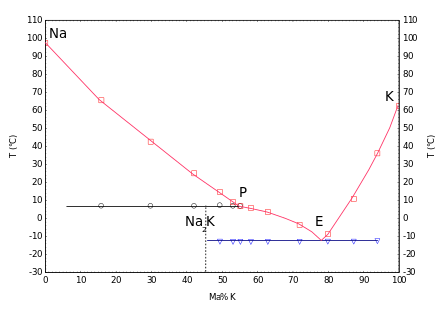
With potassium , liquid mixtures are formed in a wide range of concentrations at room temperature. The phase diagram shows an incongruent melting compound Na 2 K at 7 ° C and a eutectic at −12.6 ° C with a potassium content of 77% ( mass fraction ).
Chemical properties
Like the other alkali metals , sodium is a very base element ( normal potential : −2.71 V) and reacts easily with many other elements and sometimes with compounds. The reactions are particularly violent with non-metals such as chlorine or sulfur and take place with a bright yellow flame.
The otherwise reactive oxygen is a special feature. Sodium and oxygen do not react directly with one another without the presence of water at room temperature or when heated. In a completely anhydrous oxygen atmosphere, sodium can even be melted without reacting. On the other hand, if there are traces of moisture, it burns easily to sodium peroxide .
- Reaction of sodium with oxygen
Sodium reacts with water to form sodium hydroxide , forming hydrogen . High-speed recordings of the reaction of alkali metals with water suggest a Coulomb explosion .
- Reaction of sodium with water
In alcohols , sodium is converted into sodium alcoholates with the formation of hydrogen . It often melts due to the high heat of reaction . If the sodium is finely distributed and the resulting large reaction surface area, the reaction can be explosive and ignite the hydrogen.
- Reaction of sodium with ethanol
If sodium comes into contact with chlorinated compounds such as dichloromethane , chloroform , carbon tetrachloride , a rapid and exothermic reaction occurs with the formation of sodium chloride.

Sodium dissolves in liquid ammonia with a blue color . The color is based on free electrons that are released into the solution by the sodium. The solution also conducts electrical current and is diluted paramagnetic . The anion of sodium, the sodium ion, for example in the form of potassium (2.2.2-cryptand) natride (K + (C222) Na - ) can also be represented in a similar way . It is a very powerful reducing agent.
Isotopes
A total of 19 isotopes and 3 further core isomers from 18 Na to 37 Na are known of sodium . Of these, only one occurs naturally , the isotope 23 Na. This makes sodium one of 22 pure elements . The longest-lived artificial isotopes are 22 Na, which converts to 22 Ne with a half-life of 2.602 years under beta-plus decay (β +) and 24 Na, which also decays to 24 Mg with a half-life of 14.957 hours under beta decay. These are used as tracers in nuclear medicine . 22 Na can be produced from a cyclotron by irradiating magnesium or aluminum targets with protons for several weeks .
All other isotopes and isomers only have short half-lives of seconds or milliseconds.
use

Large amounts of sodium chloride and other sodium compounds such as sodium carbonate are pumped. But only a very small part of this is processed into sodium. Most of it is used directly or converted into other compounds. About uses of sodium compounds: see section compounds .
Sodium is the most widely used alkali metal. It is used for various purposes both technically and in the laboratory. In school lessons and during experimental lectures, sodium can be used to produce hydrogen with the aid of a sodium spoon and water. A number of sodium compounds are made from some of the sodium. These are, for example, sodium peroxide used as a bleaching agent and the strong base sodium amide . These do not occur naturally and cannot be obtained directly from sodium chloride. Sodium cyanide and sodium hydride are made from sodium. Since sodium, the solidification structure influenced, it can be used as the addition of aluminum - silicon - alloys are used (A refining process as Aladár PACZ ).
catalyst
Sodium catalyzes the polymerization of 1,3-butadiene and isoprene . Hence it was used for the production of artificial rubber . Plastic made using sodium as a catalyst, known as buna , was the world's first man-made rubber . From 1937 it was produced in the Buna works (named after butadiene and sodium) in Schkopau .
Coolant
Since sodium with a thermal conductivity of 140 W / (m · K), which is well above that of steel (15 to 58 W / (m · K)), has good heat transfer properties and also a low melting point with a large liquid range at the same time it is used as a coolant to cool the exhaust valves in internal combustion engines, which are subject to high thermal loads . For this, the valve stems are made hollow and partly filled with sodium. During operation, the sodium melts and sloshes back and forth between the hot and cold sides. The heat is transported away from the red-hot valve disc.
Fast breeders are cooled with molten sodium. In such breeder reactors, the fast neutrons produced during nuclear fission must not be slowed down between the fuel rods, as in other types of reactor . Therefore, water, which acts as a braking agent ( moderator ), must not be used for cooling . The heat is then passed on to the steam generator for turbine operation via a secondary sodium circuit.
Light generation
Sodium vapor lamps use the characteristic yellow light that sodium vapor emits during an electrical discharge. They are often used for street lighting due to their high light output.
Reducing agent
Some metals, such as titanium , zirconium , tantalum or uranium, cannot be obtained by reduction with carbon , because stable and non-separable carbides are formed. In addition to some other elements, especially aluminum and magnesium , sodium is therefore used as a reducing agent. Another element that sodium is used to make is potassium . Since potassium is a very base element, it cannot be obtained by reduction with carbon. A theoretically possible production by electrolysis is not technically possible due to the good solubility of potassium in a potassium chloride melt .
Sodium plays an important role as a reducing agent in organic synthesis . For a long time, the most important technical sodium application was the production of tetraethyl lead from chloroethane . This was an important anti-knock agent that was added to gasoline . For environmental reasons, the use of tetraethyl lead has been severely restricted or banned entirely. Therefore the consumption of sodium decreased. Otherwise, sodium is used in other reactions such as the Birch reduction and pinacol coupling . However, these are more of interest on a laboratory scale.
Desiccant
Since sodium also reacts with traces of water, freshly pressed sodium wire can be used to dry organic solvents such as diethyl ether or toluene . This method is not suitable for halogen-containing solvents (examples: methylene chloride , chloroform ) because of the violent reaction with the chlorine atom .
Sodium-potassium alloys are liquid at room temperature. These are used for heat transfer and dehalogenation in organic synthesis. Na-K is well suited for drying some well-predried solvents in order to achieve particularly low residual water contents.
Electrical conductor
During the 1960s, experiments were made with sodium cables wrapped in polyethylene . Because of the lower conductivity, a hypothetical sodium cable would have a 75% larger diameter.
| sodium | aluminum | copper | |
|---|---|---|---|
| density | 100 | 280 | 910 |
| Weight / [resistance] | 100 | 160 | 300 |
| Diameter / [resistance] | 100 | 74 | 57 |
| Price / [resistance] | 100 | 230 | 480 |
proof
The qualitative detection and the quantitative determination are carried out by atomic spectroscopy through the intense yellow color of the flame or, more precisely, through the double Na line at 588.99 nm and 589.59 nm.
The detection of sodium by purely chemical means is very difficult. Since almost all sodium compounds are readily soluble in water, classic precipitation reactions and gravimetric determinations are hardly possible. Exceptions are the yellow sodium magnesium uranyl acetate NaMg (UO 2 ) 3 (CH 3 COO) 9 · 9 H 2 O and the colorless sodium hexahydroxoantimonate Na [Sb (OH) 6 ], both of which are sparingly soluble. A precipitation reaction with the sulfate-bismuth double salt 3Na 2 SO 4 · 2Bi 2 (SO 4 ) 3 · 2H 2 O is possible. Since sodium ions are colorless in aqueous solution, color reactions are rarely carried out. In addition to ion chromatography, only spectroscopic methods are therefore of practical importance .
physiology
Sodium is one of the elements that are essential for all animal organisms . In the animal organism, sodium - together with chlorine - is the ninth most common element and is the third most common inorganic ion after calcium and potassium. Physiologically it is therefore one of the mass elements . Sodium occurs in living things in the form of Na + ions.
With an average body weight of 70 kg, the human body contains around 100 g of sodium as Na + ions. Two thirds of this is in the form of NaCl and one third is NaHCO 3 . Since it makes up 90% of the extracellular electrolytes in the human body, the sodium concentration determines the volume of the interstitial fluid via the vessel volume .
Recommended and Actual Sodium Intakes
According to the DA-CH reference values, the estimated value for the minimum intake of sodium is 550 mg / day for adults. However, various organizations have particularly recommended a maximum intake of sodium (WHO: 2 g / day; AHA: 1.5 g / day).
The actual daily sodium intake is often above these values. The reason for this is our relatively high salt consumption (2.5 g salt contains approx. 1 g sodium). The National Consumption Study II (NVS II) of the Max Rubner Institute , in which sodium consumption was determined using questionnaires, showed a median intake of 3.2 g / day (men) and 2.4 g / day (women) . The actual sodium intake is presumably even higher, as the recording via questionnaires is prone to errors. The gold standard for determining sodium intake is the determination of sodium in 24-hour urine. According to a report by the WHO, sodium excretion in the INTERSALT study was 4.1–4.5 g / day (men) and 2.7–3.5 g / day (women) in various places in Germany.
Regulation of the sodium balance
The sodium content is strictly controlled and is closely related to the regulation of the water balance . The normal concentration of sodium in serum is around 135–145 mmol / l. If the sodium level is lower, it is referred to as hyponatremia , in which there is an increase in cell volume. In hypernatremia, on the other hand, the sodium level is too high and the cells shrink. In both cases, the main impairment is the functioning of the brain. It can lead to epileptic seizures and disturbances of consciousness up to a coma . The renin-angiotensin-aldosterone system , adiuretin and atriopeptin play an important role in regulation .
The kidney is the key organ in the regulation of sodium . This is responsible for retaining water in the event of an excess of sodium in order to dilute the sodium in the body and to excrete sodium itself. If there is a sodium deficiency, more water is excreted and sodium is retained. However, it should be noted that the kidneys need some time before they can react to the changed sodium level.
Distribution in the cells
The Na + ions are not evenly distributed in the organism ; rather, as with the other ions, the concentrations inside and outside the cells are very different. These concentration gradients of Na + - and Cl - - (mainly outside), K + - as well as organic anions (mainly inside) determine the majority of the membrane potential of living cells. This membrane potential and the ion gradients are vital for most cells. Since the small inorganic ions constantly migrate to the neighboring area because of the differences in concentration, an active process is required to counteract this. The most important role is played by the sodium-potassium pump , which repeatedly pumps Na + and K + ions back while consuming energy.
Functions in nerve cells
Na + ions play an important role in the generation and transmission of excitations in nerve cells (and muscle fibers). Certain receptors are located on the postsynapses of nerve cells (and on the neuromuscular end plate of the muscle fibers) , which after activation by neurotransmitters released by the preceding nerve cell when it is excited, open and become permeable to sodium ions. The influx of sodium causes a local change in the cell's membrane potential, which is stable in the ground state. The inside becomes less negative compared to the outside, this is called a depolarization . If this depolarization is still strong enough on the way to the axon , another type of sodium channel opens. These are the voltage-dependent sodium channels of the axon, which transmit the local depolarization - together with other ion channels - through a specific opening and closing rhythm. A continuous voltage wave, the action potential, is created on the axons of the nerve cells . The sodium-potassium pump plays an essential role in restoring the basic state.
Sodium in plants
In the case of plants, however, sodium plays a subordinate role. While potassium is essential for all plants and most microorganisms, sodium is only required by some C4 and CAM plants , but usually not by C3 plants . Depending on the location, however, plants that can benefit from sodium intake have developed independently of this. These plants, called halophytes , are particularly common in coastal regions or other areas where the soil has a high concentration of sodium. Halophytes such as sugar beet , cabbage and many C4 grasses are salt-tolerant, as they can transport the sodium out of the central cylinder into the vacuoles of the leaf cells, where, as an osmotically effective ion, it increases the turgor and thereby increases cell elongation and instead of potassium positively influences leaf area growth. Sodium thus partially replaces potassium, but in another part it also has an additional effect on growth.
Plants that cannot transport sodium from the central cylinder into the leaf cells accumulate it in the xylem parenchyma . These so-called natrophobic plants include u. a. French beans and corn . The sodium, if it got into the leaf cells, could not be transported into the vacuoles, but would remain in the cell plasma ( cytosol ) and there would displace the potassium that is important for the formation of polymers (sodium-induced potassium deficiency). This ultimately led to an inhibition of photosynthesis . However, the accumulation of sodium in the central cylinder of the root and in the stem tissue has a negative effect on the plant at high sodium concentrations. The increase in the osmotic value prevents it from absorbing and transporting water. The leaves are insufficiently supplied with water and nutrients, which leads to a reduction in photosynthesis.
Since most plants contain only small amounts of sodium, many herbivores must ingest additional sodium chloride from natural salt deposits.
safety instructions
Smaller amounts of sodium are stored under petroleum . For larger quantities there are integrated handling systems with a protective gas atmosphere . Despite protective gas or petroleum, the sodium is often covered by a layer of sodium hydroxide and sodium oxide.
Sodium fires can be extinguished with metal fire powder ( table salt), potassium chloride , gray cast iron chips, or as a makeshift with sand or dry cement . However, sand and cement react to a certain extent with sodium, which reduces the extinguishing effect. Under no circumstances may water, foam, dry powder , carbon dioxide or halons be used. Some of these extinguishing agents react strongly exothermically with sodium , which can lead to stronger fires and explosions.
links
In compounds , sodium occurs exclusively in the +1 oxidation state . All compounds have a strongly ionic character, and almost all of them are readily soluble in water . Sodium compounds are among the most important salts of many acids . Sodium salts are mostly used industrially to obtain the corresponding anions , as their synthesis is inexpensive.
Halogen compounds
Sodium chloride (NaCl), often referred to as table salt or table salt, is the most important and best-known sodium salt. Since it occurs in large quantities, it is the most important raw material for the extraction of sodium and other sodium compounds. Sodium chloride is for humans the most important source of sodium is. Technically, it is, among others, to preserve food and as road salt in road use. It is named after the sodium chloride structure , a crystal structure typical of many salts .
In addition, all other possible sodium halides, i.e. sodium fluoride NaF, sodium bromide NaBr and sodium iodide NaI, are known and stable .
Oxygen compounds
A total of five oxides of sodium are known. These are sodium oxide Na 2 O, sodium peroxide Na 2 O 2 , sodium hyperoxide NaO 2 , disodium trioxide Na 2 O 3 and sodium trioxide NaO 3 . Sodium oxide is contained in many glasses ; it is created from the sodium carbonate used during glass production . When sodium is burned , it is only produced at certain temperatures (150–200 ° C) and stoichiometric amounts of sodium and oxygen . If this is not the case, sodium burns to sodium peroxide. This is a strong oxidizing agent and technically the most important sodium oxide. It is used as a bleach for textiles and paper , as well as a source of oxygen in diving and in submarines . The other oxides are very unstable and decompose quickly.
Sodium hydroxide (NaOH) is for the industry with the most important base . The aqueous solution of sodium hydroxide is called caustic soda . It is used, among other things, for the manufacture of soap and dyes and for the digestion of bauxite in aluminum production.
Sulfur compounds
With hydrogen sulfide sodium forms two salts , the sodium sulphide Na 2 S and sodium hydrosulfide NaHS. Both will u. a. used for heavy metal precipitation.
Sodium sulfate Na 2 SO 4 , the sodium salt of sulfuric acid , is u. a. Used in detergents and in the paper industry in the sulphate process . Like other divalent anions , sulfate also forms sodium hydrogen sulfate in addition to sodium sulfate . Other sulfur - oxygen acids also form sodium salts . An example is sodium thiosulfate Na 2 S 2 O 3 , which is used as a fixing salt in analog photography .
Hydrides
In sodium hydride NaH and sodium borohydride NaBH 4 , the hydrogen is in the −1 oxidation state . Both are primarily used in organic chemistry . Sodium hydride is mainly used as a strong, low nucleophilic base for the deprotonation of thiols , alcohols , amides , CH-acidic compounds, etc., whereas sodium borohydride is used for the reduction of z. B. of ketones . The latter reaction can be carried out selectively for ketones by the presence of cerium (III) compounds ( Luche reduction ). If they come into contact with water , gaseous hydrogen H 2 is produced .
More sodium compounds
Sodium carbonate Na 2 CO 3 and sodium hydrogen carbonate NaHCO 3 are the sodium salts of carbonic acid . Along with sodium chloride and sodium hydroxide , they are among the most important sodium compounds. Sodium carbonate (often referred to by the common name soda ) is used in large quantities in glass production. Sodium hydrogen carbonate is used as baking powder . When heated, it forms carbon dioxide and water with acids .
Sodium nitrate NaNO 3 , the sodium salt of nitric acid , is one of the rare naturally occurring nitrate compounds ( Chile's nitrate ). Sodium nitrate is used as a fertilizer and as a preservative .
Organic compounds of sodium, in contrast to those of lithium, are very unstable. They are extremely reactive and can sometimes react with otherwise unreactive aliphatic hydrocarbons . Only compounds with aromatic radicals, such as cyclopentadiene , which can be used as reducing agents , are sufficiently stable for applications in reactions .
Soaps are sodium or potassium salts of fatty acids . For production , fats are boiled with caustic soda or potassium hydroxide . This process is called soap boiling , the chemical reaction saponification . The fats are broken down into glycerine and the alkali salts of the fatty acids (the actual soaps). Alternatively, soaps can be made directly from free fatty acids by reacting them with alkalis to form their salts . Suitable fatty acids are, for example, lauric acid , myristic acid , palmitic acid , stearic acid , oleic acid and ricinoleic acid .
literature
- AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 .
- NN Greenwood, A. Earnshaw: Chemistry of the Elements. 1st edition. VCH Verlagsgesellschaft, Weinheim 1988, ISBN 3-527-26169-9 .
- David M. Adams: Inorganic Solids. Wiley, London 1974, ISBN 0-471-00470-7 .
Web links
- Disposal of sodium - Video about the disposal of 10 tons of sodium metal in a lake in 1947 on YouTube .
Individual evidence
- ^ Harry H. Binder: Lexicon of the chemical elements. S. Hirzel Verlag, Stuttgart 1999, ISBN 3-7776-0736-3 .
- ↑ The values for the properties (info box) are taken from www.webelements.com (sodium) , unless otherwise stated .
- ↑ Michael E. Wieser, Tyler B. Coplen: Atomic weights of the elements (IUPAC Technical Report). In: Pure and Applied Chemistry . Vol. 83, no. 2, 2011, pp. 359-396.
- ^ IUPAC, Standard Atomic Weights Revised 2013 .
- ↑ a b c d e Entry on sodium in Kramida, A., Ralchenko, Yu., Reader, J. and NIST ASD Team (2019): NIST Atomic Spectra Database (ver. 5.7.1) . Ed .: NIST , Gaithersburg, MD. doi : 10.18434 / T4W30F ( https://physics.nist.gov/asd ). Retrieved June 11, 2020.
- ↑ a b c d e Entry on sodium at WebElements, https://www.webelements.com , accessed on June 11, 2020.
- ^ NN Greenwood, A. Earnshaw: Chemistry of the elements. 1st edition. VCH, Weinheim 1988, ISBN 3-527-26169-9 , p. 97.
- ↑ Robert C. Weast (Ed.): CRC Handbook of Chemistry and Physics. CRC (Chemical Rubber Publishing Company), Boca Raton 1990, ISBN 0-8493-0470-9 , pp. E-129 to E-145. Values there are based on g / mol and given in cgs units. The value specified here is the SI value calculated from it, without a unit of measure.
- ↑ a b Yiming Zhang, Julian RG Evans, Shoufeng Yang: Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks. In: Journal of Chemical & Engineering Data . 56, 2011, pp. 328-337, doi: 10.1021 / je1011086 .
- ↑ A. Klemm, G. Hartmann, L. Lange: Sodium and Sodium Alloys. In: Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag, Weinheim 2005, doi: 10.1002 / 14356007.a24_277 .
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 84th edition. CRC Press, Boca Raton, Florida 2003.
- ↑ Ludwig Bergmann, Clemens Schaefer, Rainer Kassing: Textbook of Experimental Physics . Volume 6: Solids. 2nd Edition. Walter de Gruyter, 2005, ISBN 3-11-017485-5 , p. 361.
- ↑ a b Entry on sodium in the GESTIS substance database of the IFA , accessed on August 9, 2016(JavaScript required) .
- ↑ Entry on sodium in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ a b Mineral Atlas: Sodium .
- ↑ David Stifter: Hallstatt - In Iron Age Tradition? (PDF; 352 kB) In: Raimund Karl, Jutta Leskovar (Hrsg.): Interpretierte Eisenzeit. Case studies, methods, theory. Conference contributions from the 1st Linz Discussions on Interpretative Iron Age Archeology. (= Studies on the cultural history of Upper Austria. 18). Upper Austrian State Museum, Linz 2005, pp. 229–240.
- ^ AGW Cameron: Abundances of the elements in the solar system. In: Space Science Reviews . 15, 1970, pp. 121-146; doi: 10.1007 / BF00172440 .
- ^ KH Wedepohl: The composition of the continental crust. In: Geochimica et Cosmochimica Acta . 59, 7, 1995, pp. 1217-1232; doi: 10.1016 / 0016-7037 (95) 00038-2 .
- ↑ a b c d e f g h A. F. Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 102nd edition. Walter de Gruyter, Berlin 2007, ISBN 978-3-11-017770-1 .
- ↑ Summary on alkali metals by wiley-vch (PDF; 423 kB).
- ↑ a b K. Schubert: A model for the crystal structures of the chemical elements. In: Acta Crystallographica . B30, 1974, pp. 193-204; doi: 10.1107 / S0567740874002469 .
- ↑ Sodium at webelements.com, physical properties .
- ↑ GLCM van Rossen, H. van Bleiswijk: About the state diagram of the potassium-sodium alloys. In: Journal of Inorganic Chemistry . Volume 74, 1912, pp. 152-156; doi: 10.1002 / zaac.19120740115 .
- ↑ PE Mason, F. Uhlig, V. Vaněk, T. Buttersack, S. Bauerecker, P. Jungwirth: Coulomb explosion during the early stages of the reaction of alkali metals with water. In: Nature chemistry . Volume 7, number 3, March 2015, pp. 250-254, doi: 10.1038 / nchem.2161 . PMID 25698335 .
- ↑ DL Ward, RH Huangj, JL Dye: The Structures of Alkalides and Electrides. III. Structure of Potassium Cryptand [2.2.2] Natride. In: Acta Cryst. C46, 1990, pp. 1833-1835.
- ↑ a b c G. Audi, FG Kondev, Meng Wang, WJ Huang, S. Naimi: The NUBASE2016 evaluation of nuclear properties. In: Chinese Physics C. 41, 2017, S. 030001, doi : 10.1088 / 1674-1137 / 41/3/030001 ( full text ).
- ↑ C. Naidoo, NP van der Meulen, C Vermeulen, R. Krause-Rehberg: The Production of 22Na Positron Sources at iTHEMBA Labs. Poster (PDF)
- ↑ Wayne A. Taylor, Richard C. Heaton, David J. Jamriska: Production of sodium-22 from proton irradiated aluminum U.S. Patent 5,487,880, January 30, 1996
- ↑ stud.uni-giessen.de: Reaction of sodium with water , accessed on January 29, 2017
- ↑ K. Ziegler, F. Bersch, H. Wollthan: Investigations on organic alkaline compounds. XI. The mechanism of polymerization of unsaturated hydrocarbons by alkali metals and alkali alkyls. In: Justus Liebig's Annals of Chemistry . 511, 1, 1934, pp. 13-44; doi: 10.1002 / jlac.19345110103 .
- ^ German Chemistry Museum Merseburg, history of the Busa works in Schkopau .
- ↑ M. Volkmer: Basic knowledge of nuclear energy . Hamburgische Elektricitäts-Werke-AG, 1996, ISBN 3-925986-09-X , pp. 52-53.
- ↑ R. Brückner: reaction mechanisms. 3. Edition. Spektrum Akad. Verlag, 2004, ISBN 3-8274-1579-9 .
- ↑ Patent US3534132 : Method of making an insulated sodium cable. Filed May 9, 1967 , published October 13, 1970 , Applicant: General Electric, Inventor: Carl A. Bailey, Raymond E. Isaacson.
- ^ GF Moore (Ed.): Electric Cables Handbook. 3. Edition. Blackwell Science, 1997, ISBN 0-632-04075-0 , p. 80.
- ↑ As of August 2014 (Cu: 6600 US $ / t, Al: 2000 US $ / t, Na: 500 US $ / t).
- ↑ F. Kögler: The determination of sodium as sodium-magnesium-uranyl acetate. In: Angewandte Chemie . 48, 1935, pp. 561-565, doi: 10.1002 / anie.19350483403 .
- ↑ Science online lexica: Entry on sodium in the lexicon of chemistry .
- ↑ Entry on sodium. In: Römpp Online . Georg Thieme Verlag, accessed on September 30, 2014.
- ^ W. Kaim, B. Schwederski: Bioinorganische Chemie. 4th edition. Teubner, Wiesbaden 2005, ISBN 3-519-33505-0 .
- ↑ P. Deetjen, E.-J. Speckmann, J. Hescheler: Physiology. Elsevier, Munich 2005.
- ↑ German Society for Nutrition e. V. Reference values for nutrient intake . Retrieved December 13, 2013.
- ^ World Health Organization: Global Strategy on Diet, Physical Activity and Health. Population sodium reduction strategies . Retrieved December 13, 2013.
- ^ American Heart Association The American Heart Association's Diet and Lifestyle Recommendations . Retrieved December 13, 2013.
- ↑ Max Rubner Institute, Federal Research Institute for Nutrition and Food National Consumption Study II. Results report part 2. The nationwide survey on the nutrition of adolescents and adults. (PDF; 3.3 MB).
- ^ P. Elliott, I. Brown: Sodium intakes around the world. Background document prepared for the Forum and Technical meeting on Reducing Salt Intake in Populations (Paris 5-7th October 2006). (PDF; 2.2 MB) 2006.
- ↑ W. Siegenthaler (Ed.): Clinical Pathophysiology. 9th edition. Georg Thieme Verlag, Stuttgart 2006, ISBN 3-13-449609-7 , pp. 165-175.
- ↑ P. Deetjen, E.-J. Speckmann, J. Hescheler: Physiology. Elsevier, Munich 2005.
- ↑ a b Müller-Esterl: Biochemistry. An introduction for doctors and scientists. Spectrum Academic Publishing House, 2004, ISBN 3-8274-0534-3 .
- ↑ P. Sitte, EW Weiler u. a .: Strasburger. Textbook of botany. 35th edition. Spectrum Academic Publishing House, 2002, ISBN 3-8274-1010-X .
- ↑ K. Mengel: Nutrition and metabolism of the plant. 7th edition. Gustav Fischer Verlag, Jena 1991, ISBN 3-334-00310-8 , pp. 347-349.
- ↑ Chr. Elschenbroich: Organometallchemie. 5th edition. Teubner, 2005.
- ↑ Hans-Dieter Jakubke, Ruth Karcher (Ed.): '' Lexikon der Chemie '', Spektrum Akademischer Verlag, Heidelberg, 2001.