Sodium salts
A sodium salt is the salt of an inorganic or an organic acid . It is created by the reaction of the acid with sodium , caustic soda or the sodium salts of weaker acids.
| Sodium salts of individual fatty acids |
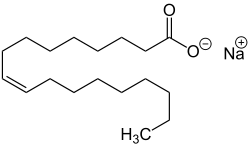
| Sodium oleate, the sodium salt of oleic acid . |
| Sodium palmitate, the sodium salt of palmitic acid . |
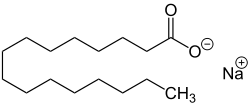
| Sodium stearate , the sodium salt of stearic acid . |
Organic sodium salts
One distinguishes between
- Sodium salts of carboxylic acids ( sodium formate , HCOONa, is the sodium salt of formic acid . Sodium acetate , CH 3 COONa, is the sodium salt of acetic acid etc.) and
- Sodium salts of sulfonic acids .
Medicinal substances
In galenics , acidic medicinal substances are often converted into sodium salts, as these are more stable, more soluble or more permeable to the membrane than the medicinal substance itself. Examples (selection) of such sodium salts are bispyribac , bithionol , bosentan , brequinar , bromfenac , cefmenoxime , ceftiofur , citicoline , diclofenac , Floxacillin , fosinopril , naproxen , netobimin , ozagrel , pantoprazole , pemetrexed , sitamaquin , sitaxentan , sulfamerazine , sulfapyridine , sulfaquinoxaline , sulfathiazole , sulfazecine , thiamylal and mesna .
Most of these salts are sodium salts of organic carboxylic acids or sulfonic acids .
The disodium salt of cromoglicic acid is also a medicinal substance, as is the disodium salt of cefodizime .
Pesticides
Herbicides are often used as sodium salts for the same reasons. An example of this is the sodium salt of flupyrsulfuron-methyl .
Cosmetics
Sodium salts of long-chain sulfonic acids (e.g. sodium lauryl sulfate ) are often found in toothpaste and shampoo . The sodium salts of fatty acids can serve as soaps and are more precisely called sodium soaps (e.g. curd soap ).
Dye manufacturing
Sodium salts of certain aromatic sulfonic acids - especially naphthalenesulfonic acids - are used in the manufacture of azo dyes .
Inorganic sodium salts
Examples of important inorganic sodium salts are sodium fluoride , sodium chloride , sodium bromide , sodium iodide , sodium sulfate , sodium hydrogen carbonate and sodium carbonate . Sodium amide (NaNH 2 ) is the sodium salt of ammonia (NH 3 ).
Some acids, e.g. B. phosphoric acid , can form three different sodium salts:
- Sodium dihydrogen phosphate (NaH 2 PO 4 , "monosodium phosphate")
- Disodium hydrogen phosphate (Na 2 HPO 4 , "disodium phosphate")
- Sodium phosphate (Na 3 PO 4 , "trisodium phosphate")
These are approved as food additives in the European Union under the common number E 339 .
See also
Individual evidence
- ^ The Merck Index . An Encyclopaedia of Chemicals, Drugs and Biologicals . 14th edition, 2006, ISBN 978-0-911910-00-1 .
- ^ The Merck Index . An Encyclopaedia of Chemicals, Drugs and Biologicals . 14th edition, 2006, p. 718, ISBN 978-0-911910-00-1 .