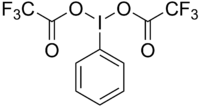
(Bis (trifluoroacetoxy) iodine) benzene
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | [Bis (trifluoroacetoxy) iodine] benzene | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 10 H 5 F 6 IO 4 | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 430.04 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
121-124 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
[Bis (trifluoroacetoxy) iodine] benzene or phenyl iodine (III) bis (trifluoroacetate) (abbreviated PIFA ) is a hypervalent iodobenzene compound. It is used as reagent in the Hofmann rearrangement in the production of amines from amides with loss of a carbon - atom used.
The reagent can also be used to prepare diazo compounds from hydrazones .
Manufacturing
As with all hypervalent iodobenzenes, the starting compound for the synthesis is iodobenzene . PIFA is made by reacting iodobenzene with a mixture of trifluoroperacetic acid and trifluoroacetic acid.
Individual evidence
- ↑ Data sheet [Bis (trifluoroacetoxy) iodo] benzene 97% from Sigma-Aldrich , accessed on January 22, 2012 ( PDF ).
- ↑ a b c data sheet bis (trifluoroacetoxy) iodobenzene (PDF) from Merck , accessed on August 31, 2011.
- ↑ Almond, MR; Stimmel, JB; Thompson, EA; Loudon, GM: Hofmann Rearrangement under mildly acidic Conditions using (I, I-bis (Trifluoroacetoxy)) iodobenzene: Cyclobutylamine Hydrochloride from Cyclobutanecarboxamide In: Organic Syntheses . 66, 1988, p. 132, doi : 10.15227 / orgsyn.066.0132 ; Coll. Vol. 8, 1993, p. 132 ( PDF ).

