1,2-bis (dimethylphosphino) ethane
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 1,2-bis (dimethylphosphino) ethane | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 6 H 16 P 2 | ||||||||||||||||||
| Brief description |
colorless liquid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 150.07 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
0.9 g cm −3 at 25 ° C |
||||||||||||||||||
| boiling point |
180 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
1,2-Bis (dimethylphosphino) ethane ( dmpe ) is a diphosphine with frequent use in coordination chemistry . It serves as a strongly basic observer ligand. Representative complexes are V (dmpe) 2 (BH 4 ) 2 , Mn (dmpe) 2 (AlH 4 ), Tc (dmpe) 2 (CO) 2 Cl, and Ni (dmpe) Cl 2 .
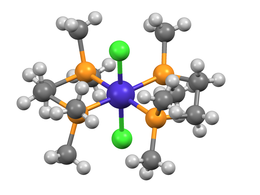
Structure of trans -CoCl 2 (dmpe) 2 ( P is colored ocher, Co blue and Cl green.)
It is a colorless, air-sensitive liquid that is soluble in organic solvents.
synthesis
The synthesis is carried out by reacting methyl magnesium iodide with 1,2-bis (dichlorophosphino) ethane:
Alternatively, dmpe can also be obtained by alkylating sodium dimethyl phosphide .
The synthesis from thiophosphoryl trichloride has led to serious accidents and should no longer be used.
Related ligands
Tetramethylethylenediamine is the diamine analog from dmpe. Bis (dicyclohexylphosphino) ethane is a sterically more demanding analogue, which also has the advantage of being a solid.
Individual evidence
- ↑ a b c d e data sheet 1,2-bis (dimethylphosphino) ethane from Sigma-Aldrich , accessed on October 13, 2019 ( PDF ).
- ↑ JE Bercaw, GW Parshall: Preparation of Tetramethyldiphosphine disulfides and Ethylenebis (Dimethylphosphine) (DMPE) . In: Inorganic Syntheses . John Wiley & Sons, Inc., Hoboken, NJ, USA 2007, ISBN 978-0-470-13254-8 , pp. 199-200 , doi : 10.1002 / 9780470132548.ch42 .
- ↑ RJ Burt, J. Chatt, W. Hussain, GJ Leigh: A convenient synthesis of 1,2-bis (dichlorophosphino) ethane, 1,2-bis (dimethylphosphino) ethane and 1,2-bis (diethylphosphino) ethane . In: J. Organomet. Chem. . 182, No. 2, 1979, pp. 203-206. doi : 10.1016 / S0022-328X (00) 94383-3 .
- ↑ JE Bercaw, GW Parshall: Preparation of Tetramethyldiphosphine Disulfide and Ethylenebis (dimethylphosphine) (dmpe) , Inorganic Syntheses 1985, vol. 23, p. 199-200, doi : 10.1002 / 9780470132548.ch42 .