Calcium nitride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
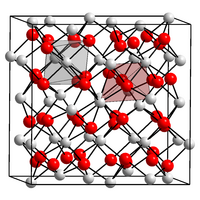
|
||||||||||||||||
| __ N 3− __ Ca 2+ | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Calcium nitride | |||||||||||||||
| Ratio formula | Ca 3 N 2 | |||||||||||||||
| Brief description |
Solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 148.25 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.63 g cm −3 (25) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Calcium nitride is an inorganic chemical compound of calcium from the group of nitrides .
Extraction and presentation
Calcium nitride can be obtained by reacting calcium (including its combustion in air) with nitrogen.
It can also be obtained by decomposing calcium amide .
properties
Calcium nitride is black to golden yellow or mixed colors, depending on the display temperature. α-calcium nitride (C-sesquioxide type or anti- bixbyite structure, space group Ia 3 (space group no. 206) with the lattice parameter a = 10.40 Å ) changes to β-calcium nitride at 700 ° C.
With water it decomposes to calcium hydroxide and ammonia .
Individual evidence
- ↑ a b c d e data sheet Calcium nitride, −200 mesh, 95% from Sigma-Aldrich , accessed on March 25, 2013 ( PDF ).
- ^ Mary Eagleson: Concise Encyclopedia Chemistry . Walter de Gruyter, 1994, ISBN 3-11-011451-8 , p. 160 ( limited preview in Google Book Search).
- ↑ a b Georg Brauer (Ed.), With the collaboration of Marianne Baudler u a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 929.
- ^ A b Richard C. Ropp: Encyclopedia of the Alkaline Earth Compounds . Newnes, 2012, ISBN 0-444-59553-8 , pp. 205 ( limited preview in Google Book search).



