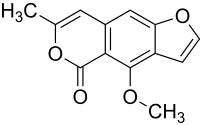
Coriandrin
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||
| General | |||||||||||||
| Surname | Coriandrin | ||||||||||||
| other names |
4-Methoxy-7-methylpyrano [3,4- f ] [1] -benzoxol-5-one |
||||||||||||
| Molecular formula | C 13 H 10 O 4 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 230.2 g · mol -1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Coriandrin is a component of the essential oils of coriander . Coriandrin was first isolated from this herb as a highly photoactive compound in 1988 and was therefore named that way. The compound is structurally derived from coumarin and belongs to the group of furanoisocoumarins.
Occurrence
The substance was found in the fruits and leaves of coriander, along with other furanocoumarins. The latter contained up to 45 µg / g leaves. There is scientifically sound evidence that naturally occurring coumarins can influence the cytochrome P450 -dependent metabolism .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Ceska, O. et al. (1988): Coriandrin, a novel highly photoactive compound isolated from Coriandrum sativum . Phytochemistry 27 (7); 2083-2087.
- ↑ Cai, Y. et al. (1993): Inhibition and inactivation of murine hepatic ethoxy- and pentoxyresorufin O-dealkylase by naturally occurring coumarins. Chemical Research in Toxicology 6; 872-879; PMID 8117927 .
