Dialkyl sulfides
| Dialkyl sulfides |
|---|
 General formula |
 Dimethyl sulfide |
 Diethyl sulfide |
In chemistry, dialkysulfides (also dialkyl thioethers ) are organic compounds that have a thioether group as a functional group - a sulfur atom that is substituted by two alkyl radicals (R 1 - S - R 2 ). They do not contain any other heteroatoms or multiple bonds . The alkyl group includes methyl , ethyl, etc. Cyclic thioethers such as. B. Tetrahydrothiophene are not included.
Dimethyl sulfide (H 3 C – S – CH 3 ) is the best-known dialkyl thioether. It has an unpleasant odor and is a component of bad breath and can also be found in traces in beer, where it is an important component of the aroma, but is perceived as unpleasant in too high concentrations. Dimethyl sulfide is released through the anaerobic breakdown of the proteinogenic amino acid methionine .
Classification
The dialkyl thioethers can be classified into symmetrical and asymmetrical, as well as straight and branched representatives.
| Ether | symmetrical | unbalanced |
|---|---|---|
| unbranched |
 Diethyl sulfide |
Ethyl methyl sulfide |
| branched |
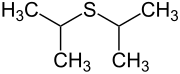 Diisopropyl sulfide |
 Ethyl isopropyl sulfide |
Manufacturing
Several methods are described for the synthesis of dialkyl sulfides:
- Reaction of thio alcoholates with haloalkanes
- Addition of thiols to alkenes
Individual evidence
- ↑ Wolfgang Legrum: Fragrances, between stink and fragrance , Vieweg + Teubner Verlag (2011), ISBN 978-3-8348-1245-2 , pp. 61-62.
- ↑ Beer mistake of the quarter: Dimethyl sulfide (DMS) homepage Braumagazin Andreas Staudt, spring 2018, accessed on February 3, 2020.
- ↑ Wolfgang Legrum: Fragrances, between stink and fragrance , Vieweg + Teubner Verlag (2011), ISBN 978-3-8348-1245-2 , pp. 78-78.
- ^ A b Siegfried Hauptmann : Organic Chemistry , 2nd revised edition, VEB Deutscher Verlag für Grundstoffindindustrie, Leipzig 1985, ISBN 3-342-00280-8 , p. 477.