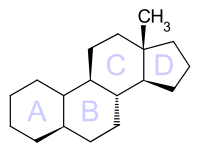
Estran
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
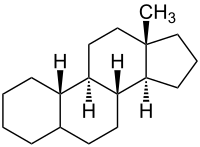
|
|||||||||||||
| General | |||||||||||||
| Surname | Estran | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 18 H 30 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 246.44 g mol −1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Estran is a polycyclic aliphatic hydrocarbon and is one of the basic components of steroids , the steranes . It consists of a tetracyclic skeleton with three fused cyclohexane rings and one fused cyclopentane ring. It differs from the Gonan in that it has an additional methyl group .
The female sex hormones ( estrogens ) are derived from estran . Examples are estrone and estradiol .
Isomers
The two cyclohexane rings A and B can be cis- or trans -linked analogously to decalin , so that the configuration isomers 5α-estrane and 5β-estrane result.
The physiologically active estrane derivatives are triply unsaturated in ring A.
Web links
Commons : Estran - collection of images, videos and audio files
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ a b entry on Estran. In: Römpp Online . Georg Thieme Verlag, accessed on June 22, 2020.
- ↑ External identifiers or database links for 5α-estran : CAS number: 517-13-5, PubChem : 6857465 , ChemSpider : 5256802 , Wikidata : Q27109326 .
- ↑ External identifiers or database links for 5β-estran : CAS number: 517-12-4, PubChem : 6857457 , ChemSpider : 5256794 , Wikidata : Q27109335 .
- ↑ Eberhard Breitmeier, Günther Jung: Organic chemistry . Basics, substance classes, reactions, concepts, molecular structure. 5th edition. Georg Thieme Verlag, Stuttgart 2005, ISBN 3-13-541505-8 , p. 944 .