Estrogens
Estrogens or estrogens (as "oestrus" -generating substances from ancient Greek οἶστρος oístrŏs , Latinized oestrus "sting", "passion" and Latin gignere "produce"), also called follicular hormones , are the most important female sex hormones from the class of steroid hormones . These are compounds that, in animal experiments, cause oestrus in castrated rodents, for example .
They are mainly produced in the ovaries (ovaries) in follicles and in the corpus luteum , and to a lesser extent in the adrenal cortex . During pregnancy, estrogens are also produced in the placenta and by the fetus . Men also produce small amounts of estrogens in their testicles ; In addition, a certain part of the testosterone in adipose tissue is converted into estrogens by an enzyme called aromatase . The amount of estrogens produced in the gonads is regulated by control hormones produced by the pituitary gland and released into the bloodstream .
Estrogens are steroids , which as a basic skeleton estran (13 β -methyl- gonane own). The first isolation of estrogens and the determination of the structure of estrogens took place in 1929 by the German chemist Adolf Butenandt .
biosynthesis
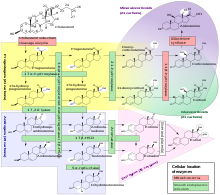
Estrogens are created by converting male sex hormones. Estradiol is mainly created by converting testosterone. The conversion is catalyzed by the enzyme aromatase . The ring A is with the C -atoms 1 to 5 and 10 of the Sterangerüstes in an aromatic structure (see aromatic ) converted.
effect
Estrogens promote the maturation of a fertile egg cell. The uterine lining is well supplied with blood by estrogens, the cervix opens, and the cervical secretion is permeable to sperm. Estrogens signal the egg cell to the pituitary gland and thus indirectly trigger ovulation. The concentration of estrogens changes significantly in the course of the female cycle . Production is controlled by the pituitary gland via the gonadotropins ( FSH and LH ). After menopause , estrogen synthesis in the female body drops sharply.
In the blood , estrogens are usually bound to protein . Specific estrogen receptors to which the estrogens bind are located on certain organs (e.g. the female breast and the uterus ) . The estrogens are transported directly to the cell nucleus and thus influence the activity of the cells. The birth control pill contains estrogens.
Estrogens promote the growth of the vagina (sheath), uterus , ovaries and fallopian tubes as well as the development of secondary female sexual characteristics such as the breasts.
Estrogens have a physiological effect on the bones in the form of epiphyseal closure and inhibition of osteoclastic bone resorption; a decrease in the level of estrogen in the blood can lead to osteoporosis (bone loss). Estrogens also have a stimulating effect on the immune system, which is why estrogen therapies occasionally activate latent autoimmune diseases as a side effect. In addition, estrogens increase the sensitivity for hearing in the brain; a reduced estrogen level, for example after menopause, worsens hearing. The hormone is essential for storing memory contents of sounds and speech.
Systemic lupus erythematosus , erythema nodosum , pruritus and erythema multiforme (all autoimmune reactions ) can occur as side effects of estrogen preparations . Estrogen can have negative effects on the psyche; the side effects of an estrogen preparation contain the information that this " very rarely <1 / 10,000 ... mental illness ... as well as insomnia and depression " can cause. Artificial sex reassignments have been observed in some animals, such as fish and frogs, under the influence of estrogens and halogenated hydrocarbons .
Estrogen types
Of the natural estrogens, only estriol and - in high doses - estradiol show an effect when administered orally. Because of their rapid metabolism, estradiol and other natural estrogens have a volatile effect when administered parenterally and only slightly when administered orally. For this reason, artificial estrogens are mainly used for medicinal purposes, which are mostly metabolized as esters of natural compounds or as substituted derivatives, and are therefore effective for longer. A natural or artificially produced form of estrogen that can be used to treat menopausal symptoms (hot flashes, vaginal changes, bone loss) or to treat a lack of ovulation and to relieve symptoms in advanced stages of prostate and breast cancer is called (with sulfuric or glucuronic acid ) called coupled estrogen .
Natural estrogens
- Estrone (E1)
- Estradiol (E2)
- Estriol (E3)
- Estetrol (E4)
- Horse estrogens (lat. Equus = horse): Equol , Equilin , Equilenin
- Fusarium estrogens ( Zea mays = maize): Zearalenone , Zeranol
Synthetic estrogens with 17 α -ethynyl group
- 57-63-6: ethinyl estradiol
- 72-33-3: Mestranol
- 13655-95-3: 11 β - methyl-ethynylestradiol
- 28913-23-7: Turisteron
- 34816-55-2: Moxestrol (11 β -methoxy-ethinylestradiol)
Other estrogens
- 2208-12-0: 6-dehydroestrone
- 53-63-4: 17 deoxyestradiol
- 362-05-0: 2-hydroxyestradiol
- 517-04-4: isoestradiol (8 α -estradiol)
- 1818-12-8: 2-methylestradiol
- 6171-48-8: 4-methylestradiol
- 28014-46-2: polyestradiol phosphate
- 39219-28-8: Promestrials
- 88847-87-4: 2-chloroestradiol
- 126559-87-3: 1.11 β- ethanoestradiol
Non-steroidal estrogens
- 56-53-1: C 18 H 20 O 2 : diethyl stilbestrol
- 84-17-3: C 18 H 18 O 2 : dienestrol
- 130-79-0: C 20 H 24 O 2 : dimestrol
- 569-57-3: C 23 H 21 ClO 3 : chlorotrianise
- 6202-26-2: C 25 H 26 O 2 : stilbestrol monobenzyl ether
- 13425-53-1: C 18 H 22 O 8 P 2 : fosfestrol
- 22059-16-1: C 19 H 26 O 2 : Homoestradiol (backbone is perhydro chrysene instead Steran )
Hyperandrogenism
About five to ten percent of all women suffer from the consequences of an uneven distribution of male and female hormones, with one cause of the so-called polycystic ovary syndrome PCOS .
See also
literature
- Lois Jovanovic, Genell J. Subak-Sharpe: Hormones. The medical manual for women. (Original edition: Hormones. The Woman's Answerbook. Atheneum, New York 1987) From the American by Margaret Auer, Kabel, Hamburg 1989, ISBN 3-8225-0100-X , pp. 65 ff., 88 ff., 121 ff., 139 ff., 148 ff., 199 ff., 204 ff., 248 ff., 258 ff., 263 ff., 348 ff. And 382.
Web links
- The two faces of estrogen . Wissenschaft.de
- Does estrogen make women's faces beautiful? (Telepolis)
- What makes women sensitive and men pain-free - testosterone dampens and estrogen increases the sensation of pain . Wissenschaft.de
Individual evidence
- ↑ Jochen Suss: Estrogens. In: Werner E. Gerabek , Bernhard D. Haage, Gundolf Keil , Wolfgang Wegner (eds.): Enzyklopädie Medizingeschichte. De Gruyter, Berlin / New York 2005, ISBN 3-11-015714-4 , pp. 1064-1066, here: p. 1064.
- ↑ Helga Satzinger, Adolf Butenandt, Hormone and Gender, in: Wolfgang Schieder, Achim Trunk, Adolf Butenandt and the Kaiser Wilhelm Society, Wallsteinverlag 2004 p. 102, cf. Schering (2) in the Sybodo Museum, Innsbruck.
- ↑ Christian Behl: Estrogen: Mystery drug for the brain? The neuroprotective activities of the female sex hormone: The Neuroprotective Activities of the Female Sex Hormone. Springer, Vienna, 2001, ISBN 3-211-83539-3 .
- ↑ Ulrich Meyer: The history of estrogens. In: Pharmazie in our time , Volume 33, 2004, No. 5, pp. 352-356. doi: 10.1002 / pauz.200400080 .
- ↑ Estrogens prepare the inner wall of the uterus for fertilization and for embedding and nourishment of the embryo at the beginning of pregnancy.
- ↑ Osteoporosis (bone loss) . In: Pschyrembel , Clinical Dictionary . 259th edition. 2002.
- ↑ Janne Kieselbach: Coronavirus: Does it hit men harder? In: Spiegel Online Wissenschaft. Accessed March 31, 2020 .
- ^ Estrogen Controls How The Brain Processes Sound , article in Science Daily of May 6, 2009.
- ↑ a b Swiss drug list: Vagifem .
- ↑ Hormone-active substances in the water . ( Memento of March 11, 2004 in the Internet Archive ; PDF) BUND, September 2001.
- ↑ Lois Jovanovic, Genell J. Subak-Sharpe: Hormones. The medical manual for women. P. 382.
- ↑ CJ Mirocha, B. Schauerhamer, CM Christensen, ML Niku-Paavola, M. Nummi: Incidence of zearalenol (Fusarium mycotoxin) in animal feed . In: Applied and Environmental Microbiology . tape 38 , no. 4 , October 1979, p. 749-750 , PMID 161492 .
- ↑ What are Fusarium toxins actually . lci-koeln