Faropenem
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
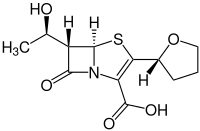
|
||||||||||||||||
| General | ||||||||||||||||
| Non-proprietary name | Faropenem | |||||||||||||||
| other names |
(+) - (5 R , 6 S ) -6 - [(1 R ) -1-Hydroxyethyl] -7-oxo-3 - [(2 R ) -tetrahydro-2-furyl] -4-thia-1- acabicyclo [3.2.0] hept-2-en-2-carboxylic acid ( IUPAC ) |
|||||||||||||||
| Molecular formula | C 12 H 15 NO 5 S | |||||||||||||||
| Brief description |
colorless to brownish powder (sodium hydrate) |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| Drug information | ||||||||||||||||
| ATC code | ||||||||||||||||
| Drug class | ||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 285.32 g · mol -1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Faropenem is an orally active β-lactam antibiotic belonging to the peneme subgroup . It was developed by Daiichi Asubio Pharma and approved in Japan in 1997 for the treatment of certain infectious diseases caused by correspondingly sensitive pathogens. In the USA, an application for approval for Faropenem medoxomil for the treatment of sinusitis , community- acquired pneumonia , acute worsening of chronic bronchitis and uncomplicated skin infections failed because of the insufficiently proven effectiveness. Faropenem is used pharmaceutically in the form of its sodium salt or the prodrug Faropenemmedoxomil.
further reading
- I. Milazzo, G. Blandino, F. Caccamo, R. Musumeci, G. Nicoletti, A. Speciale: Faropenem, a new oral penem: antibacterial activity against selected anaerobic and fastidious periodontal isolates. In: J Antimicrob Chemother . 51, No. 3, March 2003, pp. 721-725. doi: 10.1093 / jac / dkg120 . PMID 12615878 .
- IA Critchley, SD Brown, MM Traczewski, GS Tillotson, N. Janjic: National and regional assessment of antimicrobial resistance among community-acquired respiratory tract pathogens identified in a 2005-2006 US Faropenem surveillance study. In: Antimicrob Agents Chemother . 51, No. 12, December 2007, pp. 4382-4389. doi: 10.1128 / AAC.00971-07 . PMID 17908940 . PMC 2168020 (free full text).
- S. Mushtaq, R. Hope, M. Warner, DM Livermore: Activity of faropenem against cephalosporin-resistant Enterobacteriaceae. In: J Antimicrob Chemother. 59, No. 5, May 2007, pp. 1025-1030. doi: 10.1093 / jac / dkm063 . PMID 17353220 .
- JP Gettig, CW Crank, AH Philbrick: Faropenem medoxomil. In: Ann Pharmacother . 42, No. 1, January 2008, pp. 80-90. doi: 10.1345 / aph.1G232 . PMID 18094341 .
See also
Individual evidence
- ↑ a b Datasheet Faropenem sodium hydrate from Sigma-Aldrich , accessed on May 28, 2017 ( PDF ).
- ↑ Asubio Corporate History ( Memento of the original from May 17, 2012 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice. , accessed May 30, 2012.
- ↑ Schurek et al .: Faropenem: review of a new oral penem. In: Expert Review of Anti-Infective Therapy . 5, pp. 185-198, 2007, PMID 17402834 .