Fluoroperchlorate
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
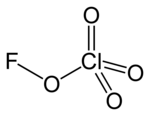
|
||||||||||
| General | ||||||||||
| Surname | Fluoroperchlorate | |||||||||
| other names |
Chlorotetroxide fluoride |
|||||||||
| Molecular formula | FClO 4 | |||||||||
| Brief description |
colorless, pungent, sour-smelling gas |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 118.46 g mol −1 | |||||||||
| Physical state |
gaseous |
|||||||||
| Melting point |
−167.3 ° C |
|||||||||
| boiling point |
−15.9 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Fluoroperchlorate is a chemical compound from the group of perchlorates .
Extraction and presentation
Fluoroperchlorate can be obtained by reacting perchloric acid with fluorine at low temperatures.
properties
Fluoroperchlorate is a colorless, strongly acidic smelling, very explosive gas. It often explodes when it melts, condenses or as a gas (similar to nitroxyfluoride NO 3 F) when it comes into contact with dust, grease or rubber. In the open test tube, the gas explodes on contact with a flame or a spark gap.
safety instructions
Fluoroperchlorate irritates the throat and lungs. It creates long-lasting shortness of breath.
Individual evidence
- ↑ a b c d e f g Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 180.
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
