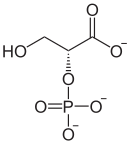
2-phosphoglyceric acid
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||
| Structural formula of ( R ) -2-phosphoglyceric acid | |||||||||||||
| General | |||||||||||||
| Surname | 2-phosphoglyceric acid | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 3 H 7 O 7 P | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 186.06 g mol −1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
2-Phosphoglyceric acid is a chemical compound from the group of phosphorylated hydroxycarboxylic acids .
Isomers
There are two stereoisomeric forms of 2-phosphoglyceric acid, the naturally occurring D -2-phosphoglyceric acid [synonym: ( R ) -2-phosphoglyceric acid] and the practically meaningless enantiomer L -2-phosphoglyceric acid.
Biological function
Of particular importance is D -2-phosphoglycerate , which is present as an anion under physiological conditions and is an intermediate in glycolysis and gluconeogenesis . The conversion to D - 3-phosphoglycerate takes place through the phosphoglycerate mutase . They also catalyze the reverse reaction . The enzyme enolase converts D -2-phosphoglycerate into phosphoenolpyruvate and water. This reaction is also reversible.
literature
- Jeremy M. Berg, Lubert Stryer et al .: Biochemistry. 5th edition. Spectrum Academic Publishing House, 2003, ISBN 3-8274-1303-6 .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.