Iridium (IV) fluoride
| Crystal structure | |||||||
|---|---|---|---|---|---|---|---|
|
|||||||
| __ Ir 4+ __ F - | |||||||
| General | |||||||
| Surname | Iridium (IV) fluoride | ||||||
| other names |
Iridium tetrafluoride |
||||||
| Ratio formula | IrF 4 | ||||||
| Brief description |
red solid |
||||||
| External identifiers / databases | |||||||
|
|||||||
| properties | |||||||
| Molar mass | 268.21 g mol −1 | ||||||
| Physical state |
firmly |
||||||
| safety instructions | |||||||
|
|||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||
Iridium (IV) fluoride is a chemical compound of iridium from the group of fluorides .
Extraction and presentation
Iridium (IV) fluoride can be obtained by reacting iridium (VI) fluoride with sulfur , metals or other halogens.
properties
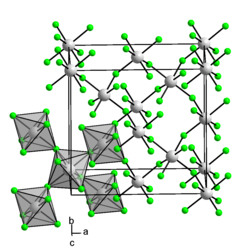
Iridium (IV) fluoride is a red solid. In humid air it decomposes, swelling and turning red. In water it hydrolyzes instantly with strong heat development and formation of iridium (IV) oxide dihydrate and hydrogen fluoride . It is reduced to lower fluorides by reducing agents when heated. It has an orthorhombic crystal structure with the space group Fdd 2 (space group no. 43) and is paramagnetic .
Individual evidence
- ↑ a b c Jane E. Macintyre: Dictionary of Inorganic Compounds . CRC Press, 1992, ISBN 978-0-412-30120-9 , pp. 3181 ( limited preview in Google Book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ a b H. J. Kandiner: Iridium . Springer-Verlag, 2013, ISBN 978-3-662-12128-3 , pp. 52 ( limited preview in Google Book search).
- ↑ PR Rao, Alain Tressaud, Neil Bartlett: The tetra fluorides of iridium, rhodium and palladium. In: Journal of Inorganic and Nuclear Chemistry. 28, 1976, p. 23, doi : 10.1016 / 0022-1902 (76) 80588-X .