Potassium titanyl phosphate
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
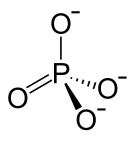 |
|||||||||||||
| General | |||||||||||||
| Surname | Potassium titanyl phosphate | ||||||||||||
| other names |
KTP |
||||||||||||
| Molecular formula | KTiOPO 4 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 197.95 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| density |
3.0 g cm −3 |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Potassium titanyl phosphate (KTP) is an ionic compound made from potassium , titanium oxide and phosphate , the crystals of which have non-linear optical properties, which makes them interesting for optics in addition to a fairly wide transparency range from 350 nm to 4400 nm. It was first synthesized by L. Ouvard in 1890.
properties
Potassium titanyl phosphate is a colorless solid which has an orthorhombic crystal structure with the space group Pna 2 1 (space group no. 33) . It decomposes from a temperature of 1150 ° C.
Applications
KTP crystals can be used for electro-optical and non-linear optical applications. A frequent application is the frequency doubling of laser light by placing a KTP crystal inside the laser resonator . The best known example is the green DPSS - laser . Here light is first generated by means of an Nd: YVO 4 or Nd: YAG laser at a wavelength of 1064 nm, which is then converted into visible green radiation with a wavelength of 532 nm by doubling the frequency. These beam sources are also known as “KTP lasers”. In quantum optics , KTP also plays a role in generating correlated photon pairs through parametric fluorescence .
Lasers with potassium titanyl phosphate are also used in medicine for pigment treatment, for example.
Individual evidence
- ↑ Data sheet on potassium titanyl phosphate
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ a b c northropgrumman.com: Potassium Titanyl Phosphate - KTP 2011, accessed July 25, 2015
- ↑ Govindhan Dhanaraj, Kullaiah Byrappa, Vishwanath Prasad, Michael Dudley: Springer Handbook of Crystal Growth . Springer Science & Business Media, 2010, ISBN 978-3-540-74761-1 , p. 719 ( limited preview in Google Book search).
- ↑ Ulf Leonhardt: Essential Quantum Optics From Quantum Measurements to Black Holes . Cambridge University Press, 2010, ISBN 978-0-521-86978-2 , pp. 160 ( limited preview in Google Book Search).
- ↑ William D. James, Timothy Berger, Dirk Elston: Andrews' Diseases of the Skin Clinical Dermatology . Elsevier Health Sciences, 2015, ISBN 0-323-31969-6 , pp. 904 ( limited preview in Google Book search).