Lithium hexafluoroantimonate (V)
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
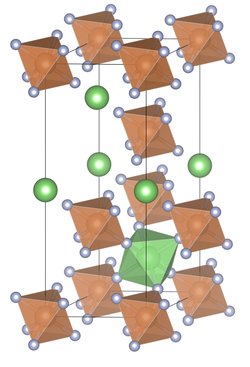
| __ Li + __ Sb 5+ __ F - | ||||||||||||||||
| Crystal system |
rhombohedral |
|||||||||||||||
| Space group |
R 3 (No. 148) |
|||||||||||||||
| Lattice parameters |
a = 5.43 Å, α = 56 ° 58 ', Z = 1 |
|||||||||||||||
| Coordination numbers |
[6] Sb, [6] Li |
|||||||||||||||
| General | ||||||||||||||||
| Surname | Lithium hexafluoroantimonate (V) | |||||||||||||||
| Ratio formula | Li [SbF 6 ] | |||||||||||||||
| Brief description |
white powder |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 242.68 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
3.2-3.5 g · cm −3 |
|||||||||||||||
| Melting point |
decomposes |
|||||||||||||||
| solubility |
soluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Lithiumhexafluoroantimonat , Li [SbF 6 ], the lithium - salt of hexafluoroantimonic H [SbF 6 ].
use
The salt is used as a photoinitiator for the production of polymers .
properties
Lithium hexafluoroantimonate is a white powder with a density between 3.2 g · cm −3 and 3.5 g · cm −3 . It is soluble in water.
The compound crystallizes in the rhombohedral space group R 3 (space group no. 148) with the lattice constants a = 5.43 Å, α = 56 ° 58 ', Z = 1. The structure corresponds to a rhombohedral distorted NaSbF 6 type.
safety instructions
The compound is toxic if swallowed or inhaled .
Combustion products of the compound are hydrogen fluoride , lithium oxide and antimony oxides .
literature
- NJ Dudney (Ed.): Battery / Energy Technology (General) - 215th ECS Meeting . Issue 25. The Electrochemical Society, 2009. ISBN 1566777674 . Pp. 15-22
Individual evidence
- ↑ a b c data sheet Lithium Hexafluoroantimonate from AmericanElements, accessed March 2, 2019.
- ↑ a b c d e f g data sheet Lithium hexafluoroantimonate from AlfaAesar, accessed on March 2, 2019 ( PDF )(JavaScript required) .
- ^ John H. Burns: The Crystal Structure of Lithium Fluoroantimonate (V) . In: Acta Crystallographica . tape 15 , 1962, pp. 1098-1101 , doi : 10.1107 / S0365110X62002935 .

