Molybdenum (III) iodide
| Crystal structure | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
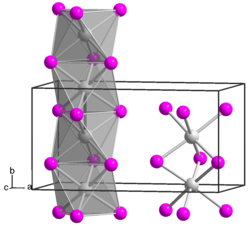
|
||||||||||
| __ Mon 3+ __ I - | ||||||||||
| General | ||||||||||
| Surname | Molybdenum (III) iodide | |||||||||
| other names |
Molybdenum triiodide |
|||||||||
| Ratio formula | MoI 3 | |||||||||
| Brief description |
black solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 476.65 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
927 ° C |
|||||||||
| solubility |
almost insoluble in water |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Molybdenum (III) iodide is an inorganic chemical compound of molybdenum from the group of iodides .
Extraction and presentation
Molybdenum (III) iodide can be produced by reacting molybdenum hexacarbonyl with iodine at 105 ° C
or molybdenum (V) chloride in a carbon disulfide solution with hydrogen iodide .
It is also possible to use molybdenum and an excess of iodine at 300 ° C.
properties
Molybdenum (III) iodide is a black, antiferromagnetic solid that is stable in air at room temperature. In a vacuum it decomposes from a temperature of 100 ° C into molybdenum (II) iodide and iodine. It is insoluble in polar and non-polar solvents . Molybdenum (III) iodide has a crystal structure isotypic to that of zirconium (III) iodide .
Individual evidence
- ↑ a b c d e f Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1539.
- ^ A b Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis US, 2011, ISBN 1-4398-1462-7 , pp. 279 ( limited preview in Google Book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ by Christoph Janiak, Hans-Jürgen Meyer, Dietrich Gudat, Ralf Alsfasser: Riedel Modern Inorganic Chemistry - Christoph Janiak, Hans-Jürgen Meyer, Dietrich Gudat, Ralf Alsfasser . Walter de Gruyter, 2012, ISBN 3-11-024901-4 , p. 357 ( limited preview in Google Book search).

