Molybdenum hexacarbonyl
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
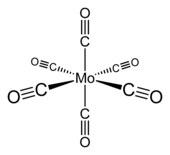
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Molybdenum hexacarbonyl | |||||||||||||||
| other names |
Molybdenum carbonyl |
|||||||||||||||
| Molecular formula | [Mo (CO) 6 ] | |||||||||||||||
| Brief description |
colorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 264.00 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.96 g cm −3 (25 ° C) |
|||||||||||||||
| Melting point |
150 ° C |
|||||||||||||||
| boiling point |
156 ° C |
|||||||||||||||
| solubility |
almost insoluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Molybdenum hexacarbonyl is a chemical compound with the formula [Mo (CO) 6 ]. Like its chromium and tungsten analogues, this colorless compound is a volatile, relatively air-stable complex of molybdenum in the zero oxidation state. Molybdenum hexacarbonyl was found in trace concentrations in the gaseous fumes from sewage sludge.
presentation
Molybdenum hexacarbonyl is represented by the reduction of molybdenum hexachloride (MoCl 6 ) under carbon monoxide pressure . It is seldom made this way in the laboratory, however, because the equipment required is expensive and the connection is inexpensive to purchase.
It can also be represented by reductive high pressure carbonylation of molybdenum (V) chloride in the presence of Devarda alloy as a halide acceptor.
Properties and structure
Molybdenum hexacarbonyl has an octahedral geometry (O h ). The six carbon monoxide ligands are positioned radially around the central molybdenum atom. The dipole moment of the complex is 0 Debye . The Mo-C distance is 206 pm . The wave number of the CO stretching vibration ν CO of the free carbon monoxide is 2004 cm −1 . It is a stable 18-valence electron complex .
The connection is relatively stable in air. It is sparingly soluble in non-polar organic solvents. Like all metal carbonyls, molybdenum hexacarbonyl is a source of volatile metal and carbon monoxide if improperly handled.
Reactions
The carbon monoxide ligands in molybdenum hexacarbonyl can be substituted by other ligands . It behaves similarly to the analog tungsten complex. Molybdenum hexacarbonyl reacts with 2,2'-bipyridine to form a heteroleptic complex Mo (CO) 4 (bipy). The UV photolysis of molybdenum hexacarbonyl in tetrahydrofuran yields a THF-substituted complex Mo (CO) 5 (THF). Many metal carbonyls can be activated photochemically in a similar manner . The thermal reaction of molybdenum hexacarbonyl with piperidine provides a complex in which two carbon monoxide ligands are replaced by piperidine Mo (CO) 4 (piperidine) 2 . The two piperidine ligands in this yellow compound are bound in a labile manner so that other ligands can be introduced under mild conditions. For example, the reaction of [Mo (CO) 4 (piperidine) 2 ] and triphenylphosphine in dichloromethane yields the cis-substituted [Mo (CO) 4 (PPh 3 ) 2 ] complex. Molybdenum hexacarbonyl is converted into the tris-acetonitrile derivative by refluxing in acetonitrile . The resulting air-sensitive compound serves as the source of the Mo (CO) 3 fragment. The reaction with allyl chloride gives [MoCl (allyl) (CO) 2 (MeCN) 2 ].
use
Molybdenum hexacarbonyl and related derivatives can be used as catalysts in organic synthesis, for example for alkyne metathesis and the Pauson-Khand reaction .
Molybdenum hexacarbonyl is used as a precursor in electron beam-induced deposition . Because it evaporates easily and breaks down in the electron beam , it provides an easily accessible source of molybdenum atoms.
Individual evidence
- ↑ a b c Christoph Elschenbroich : Organometallchemie , 6th edition, Teubner, Wiesbaden 2008, ISBN 978-3-8351-0167-8 , p. 330.
- ↑ Entry on molybdenum hexacarbonyl. In: Römpp Online . Georg Thieme Verlag, accessed on December 13, 2015.
- ↑ a b Datasheet Molybdenumhexacarbonyl, ≥99.9% trace metals basis from Sigma-Aldrich , accessed on October 24, 2011 ( PDF ).
- ↑ Entry on Molybdenum hexacarbonyl at ChemBlink , accessed on December 28, 2011.
- ↑ a b Entry on molybdenum hexacarbonyl in the GESTIS substance database of the IFA , accessed on July 23, 2016(JavaScript required) .
- ↑ J. Feldmann: Determination of Ni (CO) 4 , Fe (CO) 5 , Mo (CO) 6 , and W (CO) 6 in sewage gas by using cryotrapping gas chromatography inductively coupled plasma mass spectrometry. In: J Environ Monit , 1999, 1: pp. 33-37, doi : 10.1039 / a807277i .
- ↑ Georg Brauer (ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1634.


![{\ displaystyle \ mathrm {MoCl_ {5} \ + \ 6 \ CO \ {\ xrightarrow [{}] {Cu-Al}} \ Mo (CO) _ {6}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/054dd495cb6f40fe865291b0fb7228e26dfaee88)
