Nitrobenzonitriles
| Nitrobenzonitriles | ||||||
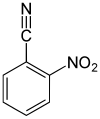
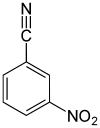
| Surname | 2-nitrobenzonitrile | 3-nitrobenzonitrile | 4-nitrobenzonitrile | |||
| other names | o -nitrobenzonitrile | m -nitrobenzonitrile | p -nitrobenzonitrile | |||
| Structural formula |
|
|

|
|||
| CAS number | 612-24-8 | 619-24-9 | 619-72-7 | |||
| PubChem | 11922 | 12079 | 12090 | |||
| Molecular formula | C 7 H 4 N 2 O 2 | |||||
| Molar mass | 148.12 g mol −1 | |||||
| Physical state | firmly | |||||
| Melting point | 108-110 ° C | 114-117 ° C | 144-147 ° C | |||
| boiling point | - | - | - | |||
|
GHS labeling |
|
|
|
|||
| H and P phrases | 302-332 | 302 + 312 + 332 | 300-311-331 | |||
| no EUH phrases | no EUH phrases | no EUH phrases | ||||
| no P-phrases | 280 | 261-264-280-301 + 310-311 | ||||
In chemistry, nitrobenzonitriles form a group of substances that is derived from both benzonitrile and nitrobenzene . The structure consists of a benzene ring with attached nitrile (–CN) and nitro group (–NO 2 ) as substituents . Their different arrangements ( ortho , meta or para ) result in three constitutional isomers with the empirical formula C 7 H 4 N 2 O 2 .
presentation
The nitrobenzonitriles can be prepared from the nitroanilines . The nitroanilines are diazotized and the diazonium salts then react according to the Sandmeyer reaction with copper (I) cyanide to give the desired product.
properties
The nitrobenzonitriles are solids. The 4-nitrobenzonitrile, which has the highest symmetry, has the highest melting point.
Individual evidence
- ↑ a b Data sheet 2-Nitrobenzonitrile from Sigma-Aldrich , accessed on March 12, 2017 ( PDF ).
- ↑ a b Data sheet 3-Nitrobenzonitrile from Sigma-Aldrich , accessed on March 12, 2017 ( PDF ).
- ↑ a b Data sheet 4-Nitrobenzonitrile from Sigma-Aldrich , accessed on May 4, 2011 ( PDF ).
- ^ Association of authors: Organikum , 19th edition, Johann Ambrosius Barth, Leipzig · Berlin · Heidelberg 1993, ISBN 3-335-00343-8 , pp. 567-568.
Web links
Commons : Nitrobenzonitrile - collection of images, videos and audio files

