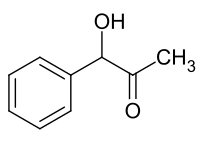
Phenylacetyl carbinol
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Phenylacetyl carbinol | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 9 H 10 O 2 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 150.17 g mol −1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Phenylacetylcarbinol (PAC) is a ketone with one phenyl group and one hydroxy group .
A catalytic reduction in the presence of methylamine converts phenylacetylcarbinol - via the intermediate stage of the imine - to ephedrine .
synthesis
Phenylacetylcarbinol is produced microbiologically from the starting materials benzaldehyde and acetaldehyde .
PAC can also consider the factors thiamine and magnesium - ions from the reaction of pyruvic acid and benzaldehyde win. Carbon dioxide is produced as a by-product .
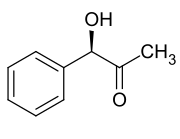
Stereoisomers
| Phenylacetyl carbinol | ||||
| Surname | (R) -phenylacetylcarbinol | (S) -Phenylacetylcarbinol | ||
| Other name |
L - (-) - phenylacetylcarbinol
(1 R ) -1-Hydroxy-1-phenyl-propan-2-one |
D - (-) - Phenylacetylcarbinol
(1 S ) -1-Hydroxy-1-phenyl-propan-2-one |
||
| Structural formula |
|

|
||
| CAS number | 1798-60-3 | 53439-91-1 | ||
| PubChem | 9920426 | 9815225 | ||
| ECHA info card | 100,015,715 | |||
| EC number | 217-285-4 | |||
literature
- PF Smith, D. Hendlin: Further studies on phenylacetylcarbinol synthesis by yeast. In: Applied Microbiology . Volume 2, Number 5, September 1954, pp. 294-296, PMID 13208181 , PMC 1057016 (free full text).
- Purer PAC production from yeast (English; PDF file; 1.17 MB)
Individual evidence
- ↑ There is not yet a harmonized classification for this substance . A labeling of 1-hydroxy-1-phenylacetone in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on July 10, 2020, is reproduced from a self-classification by the distributor .



