Xylidine Ponceau
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
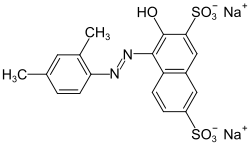
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Xylidine Ponceau | ||||||||||||||||||
| other names | |||||||||||||||||||
| Molecular formula | C 18 H 14 N 2 Na 2 O 7 S 2 | ||||||||||||||||||
| Brief description |
Dark red to burgundy red powder |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 480.42 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
> 300 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Xylidin-Ponceau is a red azo dye from the group of acid dyes that was previously used to dye wool. The dye is used in Masson-Goldner trichrome staining .
The synthesis of xylidine-Ponceau takes place by diazotization of 2,4-xylidine and azo coupling with 3-hydroxynaphthalene-2,7-disulfonic acid (R-acid).
Individual evidence
- ↑ a b c Entry on ACID RED 26 at ChemicalBook , accessed November 22, 2019.
- ↑ a b Datasheet Ponceau Xylidine, Dye content ≥60% from Sigma-Aldrich , accessed on November 4, 2013 ( PDF ).
- ^ Rudolf Nietzki: Chemistry of organic dyes . 5th edition. Julius Springer Verlag, Berlin 1906, p. 64 ff . ( limited preview in Google Book search).
