Sugammadex
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
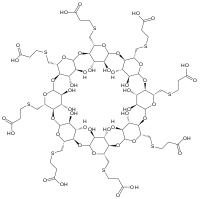
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Sugammadex | |||||||||||||||||||||
| other names | ||||||||||||||||||||||
| Molecular formula | C 72 H 112 O 48 S 8 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class |
Muscle relaxant reversor |
|||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 2002.15 g · mol -1 | |||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Sugammadex (trade name Bridion , Manufacturer: MSD Sharp & Dohme ) is a drug that the neuromuscular blocking (muscle relaxation) by muscle relaxants from the aminosteroid type, in particular rocuronium , after anesthesia (cancels reversed ). The drug has been approved by the European Commission since the end of July 2008 .
construction
Sugammadex is a modified γ- cyclodextrin with a hydrophilic outside and a lipophilic inside. The binding of negatively charged carboxylate groups creates a very stable inclusion compound, which is marketed under the catchphrase “encapsulation”. Sugammadex has a high affinity for rocuronium, for which it was developed, and a lower affinity for vecuronium and pancuronium . Non-steroidal muscle relaxants ( benzylisoquinolines such as mivacurium or atracurium ) and suxamethonium cannot be bound by sugammadex.
Mechanism of action
After intravenous administration of sugammadex, the free muscle relaxant is "encapsulated". This creates a concentration gradient for the relaxant from the neuromuscular endplate into the intravascular space , so that more secondary follows and is bound. This process stops the muscle relaxant from working. In contrast to the previously available muscle relaxant antagonists ( acetylcholinesterase inhibitors such as neostigmine ), the effect does not take place on the neuromuscular endplate.
In contrast to the preparations available to date, sugammadex is also able to clear deep neuromuscular blockages immediately after administration of rocuronium within two to three minutes, depending on the dose. The risk of recurarization (resumption of muscle relaxation) does not seem to exist; at any rate, recurarization was not observed in clinical studies with adequate sugammadex dosages.
Side effects
Sugammadex turned out to be relatively low in side effects in the approval studies. Due to its mechanism of action, sugammadex has almost no cholinergic side effects that the acetylcholinesterase inhibitors currently used have ( bradycardia , bronchoconstriction , salivation). Allergic reactions ( urticaria ) have only been observed in isolated cases .
The most common side effect is a taste disturbance (metallic or bitter), which occurs in more than 10% of patients. It is unclear whether the intraoperative wakefulness observed in some cases of use is due to sugammadex.
Web links
- Public Assessment Report (EPAR) of the European Medicines Agency (EMA) for: Sugammadex
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
literature
- WT Nicholson, J. Sprung, CJ Jankowski: Sugammadex: a novel agent for the reversal of neuromuscular blockade. In: Pharmacotherapy . 27 (8), 2007 Aug, pp. 1181-1188. PMID 17655516 .
- M. Naguib: Sugammadex: Another milestone in clinical neuromuscular pharmacology. In: Anesth Analg. 104 (3), 2007 Mar, pp. 575-581. PMID 17312211 .
- A. Bom et al .: A novel concept of reversing neuromuscular block: chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. In: Angew Chem Int Ed Engl. 41 (2), 2002 Jan 18, pp. 266-270. PMID 12491405 .
- HD De Boer et al .: Sugammadex, a new reversal agent for neuromuscular block induced by rocuronium in the anesthesized Rhesus monkey. In: Br J Anaesth. 96 (4), 2006, pp. 473-479. PMID 16464982 .
