Suvorexant
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
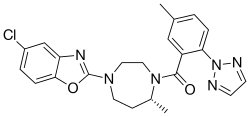
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Suvorexant | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 23 H 23 ClN 6 O 2 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| Mechanism of action |
Orexin receptor antagonist |
|||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 450.92 g mol −1 | |||||||||||||||||||||
| solubility |
|
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Suvorexant is a drug from the group of orexin receptor antagonists . It was approved in the United States in 2014 as belsomra for the treatment of sleep disorders.
The product was developed by Merck Sharp & Dohme (MSD) and is the first active ingredient from this class to be used therapeutically. Further approvals exist in Canada, Japan and Australia.
Working principle
It is believed that Suvorexant competitively and reversibly both the orexin receptors subtypes of OX 1 and OX 2 inhibits ( d ualer O Rexin r ezeptor- A ntagonist, DORA ). This neutralizes the awakening effect of the neuropeptides orexin A and B in the brain. In humans, orexinergic neurons are located in the lateral hypothalamus and from there also influence other areas of the brain that regulate the metabolism and sleep-wake rhythm .
Suvorexant is effective orally and is given as a film-coated tablet.
Indications
Suvorexant is indicated for the treatment of insomnia (difficulty falling asleep and / or staying asleep).
Contraindications
Suvorexant must not be used if you have narcolepsy .
Side effects
Loss of orexin neurons has been reported in people with narcolepsy. There is evidence that the active ingredient leads to daytime sleepiness and suicidal thoughts .
Individual evidence
- ↑ a b c caymanchem: Suvorexant , accessed April 14, 2020.
- ↑ a b c d BELSOMRA - suvorexant tablet, film coated (Prescription Information) . FDA . Status: August 2014.
- ↑ CDER New Molecular Entity (NME) and Biologic Approvals Calendar Year 2014 . FDA .
- ↑ a b T. Bennett et al .: Suvorexant, a dual orexin receptor antagonist for the management of insomnia . Pharmacy & Therapeutics, Volume 39 (2014), pp. 264-266. PMC 3989084 (free full text).
- ↑ Suvorexant: First Global Approval. In: Drugs. 2014, doi : 10.1007 / s40265-014-0294-5 . PMID 25227290 .
- ↑ BELSOMRA suvorexant - (Australian Product Information) . TGA . Status: October 2019.
- ↑ K. Gräfe: The ideal sleeping pill is still being sought , Pharmazeutische Zeitung , September 26, 2019.
