Tritoqualine
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
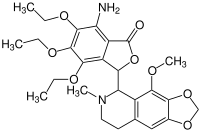
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Tritoqualine | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 26 H 32 N 2 O 8 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 500.54 g mol −1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Tritoqualine is a drug that inhibits the enzyme histidine decarboxylase and is therefore one of the atypical antihistamines . However, this does not have a sedating effect and also has an inhibitory effect on mast cell degranulation. It can be used as a support for the treatment of hives , allergic rhinitis and acute, chronic eczema as well as allergic asthmoid conditions. The drug is contraindicated in the first trimester and in type 1 diabetes and is currently out of trade in Germany .
Trade names
Inhibostamin (D, except for trade )
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ "The antiallergic Tritoqualin does not sedate" on the website of the Deutsches Ärzteblatt
- ↑ a b Pharmacology and Toxicology: for study and practice; with 281 tables . 6., completely revised and exp. Schattauer, Stuttgart New York 2007, ISBN 978-3-7945-2295-8 , pp. 354 ( dnb.de [accessed on March 29, 2017]).
- ↑ Randomized, placebo controlled study of tritoqualine (hypostamine *) in the treatment of perennial allergic rhinitis . A. Pradalier, V. Hentschel, S. Prouzeau, D. Legallais, G. Lefrançois. Revue Française d'Allergologie et d'Immunologie Clinique 43: 175-179 (2003).
- ↑ a b www.epgonline.org
- ↑ “For the time being, no Inhibostamin® for type I diabetes!” On the website of the Deutsches Ärzteblatt