Vanadium nitride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
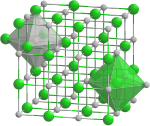
|
||||||||||||||||
| __ V 3+ __ N 3− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Vanadium nitride | |||||||||||||||
| other names |
|
|||||||||||||||
| Ratio formula | VN | |||||||||||||||
| Brief description |
black solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 65.0 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
6.04 g cm −3 |
|||||||||||||||
| Melting point |
2050 ° C |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Vanadium nitride is an intermetallic compound of vanadium from the group of nitrides .
Extraction and presentation
Vanadium nitride can be obtained by reacting vanadium with nitrogen or ammonia .
properties
Vanadium nitride is a black semi-metallic solid with a homogeneity area VN 1.0 -VN 0.7 and a crystal structure of the sodium chloride type. Vanadium nitride, vanadium carbide and vanadium (II) oxide have isotypic crystal structures in which nitrogen, carbon and oxygen can be exchanged and form mixed crystals. Vanadium nitride forms solid solutions with titanium nitride , niobium nitride , titanium carbide , niobium carbide and tantalum carbide .
Individual evidence
- ↑ a b c d e Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1436.
- ^ Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis US, 2011, ISBN 1-4398-1462-7 , pp. 451 ( limited preview in Google Book search).
- ^ A b Hugh O. Pierson: Handbook of Refractory Carbides & Nitrides: Properties, Characteristics ... William Andrew, 1996, ISBN 0-8155-1770-X , p. 200 ( limited preview in Google Book Search).
- ↑ a b Data sheet vanadium nitride, −325 mesh, 99% from Sigma-Aldrich , accessed on June 16, 2013 ( PDF ).
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Properties of the Elements and Inorganic Compounds, pp. 4-98.


