1,4-cyclohexanedione
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
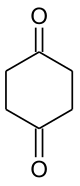
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 1,4-cyclohexanedione | |||||||||||||||
| other names |
Cyclohexane-1,4-dione |
|||||||||||||||
| Molecular formula | C 6 H 8 O 2 | |||||||||||||||
| Brief description |
light yellow odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 112.13 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
77-78 ° C |
|||||||||||||||
| boiling point |
132 ° C (20 mmHg ) |
|||||||||||||||
| solubility | ||||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
1,4-Cyclohexanedione is a chemical compound from the group of cyclanhexanedione .
Occurrence
1,4-Cyclohexanedione is found in tobacco smoke .
Extraction and presentation
1,4-Cyclohexanedione can be prepared in two steps from diesters of succinic acid. For example, the diethyl ester condenses under basic conditions to give 2,5-dicarbethoxy-1,4-cyclohexanedione. This intermediate can be hydrolyzed and decarboxylated to give the desired dione.
properties
1,4-Cyclohexanedione is a light yellow, odorless solid that is soluble in water, methanol and ethanol. It decomposes at temperatures above 190 ° C.
use
1,4-Cyclohexanedione is used in the manufacture of 1,4-benzoquinone and organic bromine. It plays an important role in the synthesis of drugs and other chemical compounds.
Individual evidence
- ↑ a b c d e f g h i data sheet 1,4-Cyclohexanedione, 98% from AlfaAesar, accessed on April 20, 2019 ( PDF )(JavaScript required) .
- ↑ Alan Rodgman, Thomas A. Perfetti: The Chemical Components of Tobacco and Tobacco Smoke, Second Edition . CRC Press, 2013, ISBN 978-1-4665-1548-2 , pp. 1799 ( limited preview in Google Book Search).
- ↑ unknown: 1,4-CYCLOHEXANEDIONE. In: Organic Syntheses. 45, 1965, p. 25, doi : 10.15227 / orgsyn.045.0025 .