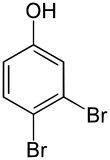
3,4-dibromophenol
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 3,4-dibromophenol | |||||||||||||||
| Molecular formula | C 6 H 4 Br 2 O | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 251.9 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
80 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
3,4-Dibromophenol is a chemical compound that belongs to both phenols and halogen aromatic compounds .
presentation
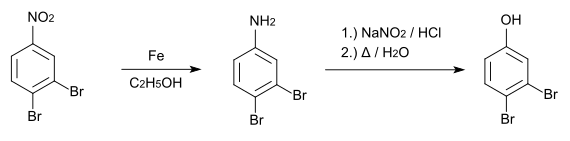
The synthesis of 3,4-dibromophenol starts from 3,4-dibromonitrobenzene , which is reduced with iron in ethanol to 3,4-dibromaniline . This is then converted to the desired compound by boiling the diazonium compound formed with sodium nitrite .
Derivatives
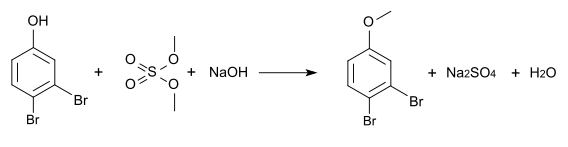
The methyl ether can be produced by methylation with dimethyl sulfate and is also known under the common name 3,4-dibromanisole (CAS number: 62415-74-1).
Individual evidence
- ^ Dictionary of organic compounds, p. 1971 ( limited preview in Google book search).
- ↑ a b Brita Liedholm, Leif K. Sydnes, Tyge Greibrokk, Torbjörn Norin, Lars Mörch: Copper (I) -induced Bromine-Hydrogen Exchange of 2,3-Dibromoanilines. In: Acta Chem. Scand., Ser. B , 1984 , 38 B, p. 877; doi : 10.3891 / acta.chem.scand.38b-0877 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.