3,4-dinitrocatechol
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||
| General | |||||||||||||
| Surname | 3,4-dinitrocatechol | ||||||||||||
| other names |
|
||||||||||||
| Molecular formula | C 6 H 4 N 2 O 6 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 200.11 g · mol -1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| Melting point |
|
||||||||||||
| pK s value |
|
||||||||||||
| solubility | |||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
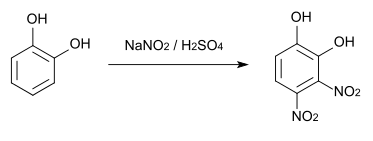
3,4-Dinitrocatechol is a chemical compound that belongs to both the phenol group and the aromatic nitro group. 3,4-dinitrocatechol crystallizes as a dihydrate.
presentation
3,4-Dinitrocatechol can be made from catechol by reacting with sodium nitrite and sulfuric acid.
Derivatives
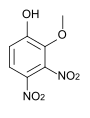
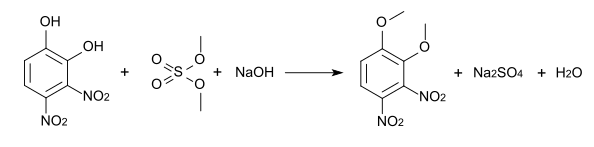
The methylation of 3,4-dinitropyrocatechol leads to different ethers, the melting points of which are listed in the table below.
use
3,4-Dinitrocatechol is used in the photometric determination of vanadium .
Individual evidence
- ↑ a b c d e f g J. Buckingham: Dictionary of organic compounds , Volume 9, p. 2767. ( limited preview in the Google book search)
- ↑ a b c d D. H. Rosenblatt, J. Epstein, M. Levitch: Some Nuclearly Substituted Catechols and their Acid Dissociation Constants , in: J. Am. Chem. Soc. , 1953 , 75 (13), pp. 3277-3278; doi : 10.1021 / ja01109a511 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.