2,6-dimethyl-4-nitrophenol
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
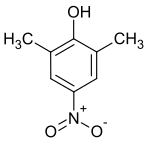
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 2,6-dimethyl-4-nitrophenol | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 8 H 9 NO 3 | ||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 167.16 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
168 ° C (dec.) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
2,6-Dimethyl-4-nitrophenol (also 4-nitro-2,6-dimethylphenol ) is an aromatic compound ; it is a phenol derivative with two methyl groups and one nitro group as substituents.
In the form of its phenolate , it is used for the photometric determination of nitrogen oxides ( dimethyphenol method ). Here, NO and NO 2 are oxidized to dinitrogen pentoxide by means of ozone , which is hydrolyzed to nitric acid in water . In sulfuric acid or phosphoric acid solution, 2,6-dimethyl-4-nitrophenol is formed with 2,6-dimethylphenol . This is now determined photometrically in the form of its phenolate.
See also
Web links
- Entry to phenol, 2,6-dimethyl-4-nitro- . In: P. J. Linstrom, W. G. Mallard (Eds.): NIST Chemistry WebBook, NIST Standard Reference Database Number 69 . National Institute of Standards and Technology , Gaithersburg MD, accessed January 4, 2020.
Individual evidence
- ↑ a b data sheet 2,6-dimethyl-4-nitrophenol from Sigma-Aldrich , accessed on January 4, 2020 ( PDF ).
- ↑ Klaus Görner, Kurt Hübner: Gas cleaning and air pollution control , Berlin / Heidelberg 2002, ISBN 3-540-42006-1 , pp. M10 – M11 ( limited preview in the Google book search).