Cytidine triphosphate
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
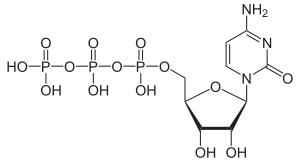
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Surname | Cytidine triphosphate | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 9 H 16 N 3 O 14 P 3 | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 483.16 g mol −1 | |||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Cytidine triphosphate ( CTP ) is a nucleoside triphosphate with the base cytosine . It is an RNA building block that is incorporated into the RNA by splitting off two phosphate groups .
CTP can bind phosphocholine by splitting off pyrophosphate and then transfer it to another molecule, e.g. B. on a diglyceride . This creates a phosphatide . Accordingly, CTP and CDP-choline (cytidine diphosphate choline) play a role in phosphatide synthesis. CTP also inhibits aspartate transcarbamoylase in pyrimidine biosynthesis .
Other ribonucleoside triphosphates are adenosine triphosphate (ATP), guanosine triphosphate (GTP) and uridine triphosphate (UTP).
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Jeremy M. Berg, Lubert Stryer, John L. Tymoczko: Stryer Biochemistry. ISBN 3827429889 p. 292 ( limited preview in Google Book Search).