Dinitrobenzoic acids
The dinitrobenzoic acids form a group of aromatic compounds that are derived from benzoic acid as well as from nitrobenzene or dinitrobenzene . The structure consists of a benzene ring with an attached carboxy group (-COOH) and two nitro groups (-NO 2 ) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 7 H 4 N 2 O 6 . The 3,5-dinitrobenzoic acid is mainly used in the analysis of organic substances by derivatization .
presentation
2,4-Dinitrotoluene is oxidized to 2,4-Dinitrobenzoic acid with sodium dichromate in sulfuric acid. From 4-nitrobenzoic acid is produced by nitrating a mixture of 2,4- and 3,4-dinitro benzoic acid. 2,6-Dinitrotoluene is metabolized to 2,6-Dinitrobenzoic acid. Oxidation of 3,4-dinitrotoluene with an ozone-oxygen mixture in the presence of cobalt (II) acetate leads to 3,4-dinitrobenzoic acid.
properties
The dinitrobenzoic acids are yellowish crystalline solids with melting points between 166 and 206 ° C. The solubilities in water vary between 18.5 g / l and poor solubility. Due to the −M effect of the two nitro groups, the dinitrobenzoic acids have a higher acidity than the benzoic acid and the nitrobenzoic acids . The pK s values are therefore correspondingly lower (benzoic acid: 4.20).
| Dinitrobenzoic acids | ||||||||||||
| Surname | 2,3-dinitrobenzoic acid |
2,4-dinitrobenzoic acid |
2,5-dinitrobenzoic acid |
2,6-dinitrobenzoic acid |
3,4-dinitrobenzoic acid |
3,5-dinitrobenzoic acid |
||||||
| Structural formula |

|
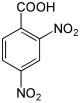
|

|

|

|
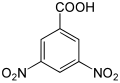
|
||||||
| CAS number | 15147-64-5 | 610-30-0 | 610-28-6 | 603-12-3 | 528-45-0 | 99-34-3 | ||||||
| PubChem | 283327 | 11880 | 11879 | 119206 | 10709 | 7433 | ||||||
| Molecular formula | C 7 H 4 N 2 O 6 | |||||||||||
| Molar mass | 212.12 g mol −1 | |||||||||||
| Physical state | firmly | |||||||||||
| Brief description | yellowish crystalline solids | |||||||||||
| Melting point | 201 ° C | 179-183 ° C | 178-182 ° C | 202 ° C (dec.) | 166 ° C | 204-206 ° C | ||||||
| pK s value (25 ° C) | 1.85 | 1.42 | 1.62 | 1.14 | 2.82 | 2.82 | ||||||
| solubility | 18.5 g / l | 6.73 g / l (25 ° C) | ||||||||||
|
GHS labeling |
|
|
|
|
|
|
||||||
| H and P phrases | see above | 315-319-335 | see above | no H-phrases | 315-319-335 | 302-315-319-335-413 | ||||||
| see above | no EUH phrases | see above | no EUH phrases | no EUH phrases | no EUH phrases | |||||||
| see above |
261-302 + 352 305 + 351 + 338-321-405-501 |
see above | no P-phrases | 261-305 + 351 + 338 | 261-305 + 351 + 338 | |||||||
Individual evidence
- ^ A b Streitwieser / Heathcock: Organische Chemie , 1st edition, Verlag Chemie, Weinheim 1980, ISBN 3-527-25810-8 , p. 1042.
- ↑ M. Mori, T. Kawajiri, M. Sayama, Y. Taniuchi, T. Miyahara, H. Kozuka: "Metabolism of 2,6-dinitrotoluene in male Wistar rat", in: Xenobiotica , 1989 , 19 (7), Pp. 731-741 ( PMID 2773509 ).
- ↑ P. Yu. Andreev, EV Potapenko, IP Pogorelova, SN Krasil'nikov, AN Volume: “A New Procedure for Preparing 3,4-Dinitrobenzoic Acid”, in: Zhurnal Prikladnoi Khimii , 2008 , 81 (3), pp. 518-520 (translated in: Russian Journal of Applied Chemistry , 81 (3), pp. 503-505, doi : 10.1134 / S1070427208030348 ).
- ↑ a b c d e f g CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
- ^ A b International Critical Tables of Numerical Data, Physics, Chemistry and Technology , Volume 1, Callie Hull, National Academies, 1926, p. 206 ( limited preview in Google Book Search).
- ↑ a b Entry on 2,4-dinitrobenzoic acid in the GESTIS substance database of the IFA , accessed on October 21, 2012(JavaScript required) .
- ↑ a b Data sheet 2,5-Dinitrobenzoic acid from Sigma-Aldrich , accessed on April 29, 2011 ( PDF ).
- ↑ a b Entry on 3,4-dinitrobenzoic acid in the GESTIS substance database of the IFA , accessed on October 21, 2012(JavaScript required) .
- ↑ a b Data sheet 3,5-Dinitrobenzoic acid from Sigma-Aldrich , accessed on December 16, 2013 ( PDF ).
