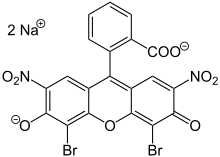
Eosin B
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Eosin B | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 20 H 6 Br 2 N 2 Na 2 O 9 | |||||||||||||||
| Brief description |
brownish green solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 624.09 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| solubility |
good in water (300 g l −1 at 20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Eosin B is a brownish green dye from the group of xanthene dyes and triphenylmethane dyes . It can be used to dye textiles and paper. In addition to eosin B, eosin Y is also known, which has a wider range of applications than eosin B. Eosin Y has the same basic structure as eosin B, but the nitro groups have been replaced by bromine atoms. The structure of Eosin B is derived from fluorescein .
presentation
The reaction of eosin Y with nitric acid can be used to synthesize eosin B. Here, two bromine atoms are exchanged for nitro groups.
use
Eosin B can be used for hematoxylin-eosin staining in histology and clinical cytology . In preparations, the cytoplasm , collagen and elastin stained with eosin B appear red, while erythrocytes appear red-orange.
Web links
Individual evidence
- ↑ a b c d Eosin B data sheet (PDF) from Merck , accessed December 9, 2010.
- ↑ A. Baeyer : About the compounds of phthalic acid with phenols , in: Liebigs Ann. , 1876 , 183 , pp. 1-74; doi : 10.1002 / jlac.18761830102 .
- ↑ Merck KGaA : Hematoxylin-Eosin Staining ( Memento from May 18, 2015 in the Internet Archive ).