Potassium pertechnetate
| Crystal structure | |||||||
|---|---|---|---|---|---|---|---|
|
|||||||
| __ K + __ Tc 7+ __ O 2− | |||||||
| Crystal system | |||||||
| Space group |
I 4 1 / a |
||||||
| Lattice parameters |
|
||||||
| General | |||||||
| Surname | Potassium pertechnetate | ||||||
| Ratio formula | KTcO 4 | ||||||
| Brief description |
white solid |
||||||
| External identifiers / databases | |||||||
|
|||||||
| properties | |||||||
| Molar mass | 201.1 g mol −1 | ||||||
| Physical state |
firmly |
||||||
| Melting point |
540 ° C |
||||||
| solubility |
2.13 g l −1 (25 ° C) |
||||||
| Hazard and safety information | |||||||
 Radioactive |
|||||||
|
|||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||
Potassium pertechnetate, KTcO 4 is a chemical compound of potassium from the group of pertechnetates .
Extraction and presentation
Kaliumpertechnetat can be prepared by the neutralization of the pertechnetic acid with potassium hydroxide can be obtained.
properties
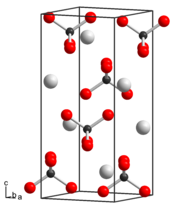
The distance of the Tc – O bond is 173.9 pm and the angle of the O – Tc – O bond is 108.05 ° and 110.19 °. The distance between the potassium and the oxygen is 289.36 pm and 286 pm. Potassium pertechnetate crystallizes in a crystal structure similar to potassium perrhenate with the space group I 4 1 / a (space group no. 88). The lattice parameters are a = 563.0 pm and c = 1286.7 pm.
Individual evidence
- ↑ Jane E. Macintyre: Dictionary of Inorganic Compounds . CRC Press, 1992, ISBN 0-412-30120-2 , pp. 3535 .
- ↑ a b c Klaus Schwochau: Technetium: Chemistry and Radiopharmaceutical Applications . John Wiley & Sons, 2008, ISBN 3-527-61337-4 , pp. 129 .
- ↑ The hazards emanating from radioactivity do not belong to the properties to be classified according to the GHS labeling. With regard to other hazards, this substance has either not yet been classified or a reliable and citable source has not yet been found.
- ^ A b Jamie Weaver, Chuck Z. Soderquist, Nancy M. Washton, Andrew S. Lipton, Paul L. Gassman, Wayne W. Lukens, Albert A. Kruger, Nathalie A. Wall, John S. McCloy: Chemical Trends in Solid Alkali pertechnetates . Inorg. Chem., February 2017, p. 2533-2544 , doi : 10.1021 / acs.inorgchem.6b02694 .
