Lithium bis (oxalato) borate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
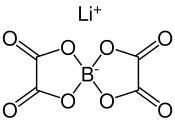
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Lithium bis (oxalato) borate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | LiB [C 2 O 4 ] 2 | |||||||||||||||
| Brief description |
White dust |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 193.79 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
290 ° C (decomposition) |
|||||||||||||||
| solubility |
soluble in propylene carbonate , ketones and lactones |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Lithium bis (oxalato) borate , or LiBOB for short , is an inorganic - chemical compound of lithium from the group of borates . LiBOB is a white solid in powder form that is soluble in anhydrous solvents such as propylene carbonate, it slowly disintegrates on contact with water.
Lithium bis (oxalato) borate is used in the electrolytes of lithium-ion batteries and can replace fluorine-containing substances such as lithium tetrafluoroborate or lithium hexafluorophosphate in this area of application . The replacement as an electrolyte in lithium-ion batteries offers the advantage of avoiding the formation of hydrogen fluoride (HF) as a highly corrosive decomposition product in the event of damage and fire on the battery.
Individual evidence
- ↑ a b c d e Lithium bis (oxalato) borate - LiBOB. Rockwood Lithium, archived from the original on December 29, 2016 ; accessed on December 29, 2016 .
- ↑ a b c d data sheet lithium bis (oxalato) borate from Sigma-Aldrich , accessed on December 30, 2016 ( PDF ).
