Methylsilanetriol
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
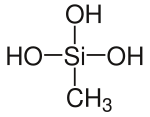
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Methylsilanetriol | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | CH 6 O 3 Si | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 94.14 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Methylsilanetriol is a chemical compound and belongs to the group of silanols . It is a derivative of silane and the silicon analogue of the unstable 1,1,1-ethanetriol.
Presentation and extraction
During the hydrolysis of trichloromethylsilane - under suitable conditions - methylsilanetriol is formed.
properties
Methylsilanetriol is a liquid and volatile organic compound .
It can be broken down into hydroxymethylsilanetriol .
use
Methylsilanetriol is a possible hydrolysis product of branch points on PDMS chains. The potassium salt ( potassium methyl siliconate, CAS number: 31795-24-1) is used as a water repellent for building materials.
See also
Web links
- EFSA : Monomethylsilanetriol added to food supplements for nutritional purposes: Scientific panel on food additives and nutrient sources added to food
- Structure and vibrational spectra of dimethylsilanediol and methylsilanetriol dimers; doi : 10.1016 / j.cplett.2005.07.018
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Entry on methylsilanetriol in the Biocatalysis / Biodegradation Database
- ↑ Entry on potassium methylsilanetriolate in the GESTIS substance database of the IFA , accessed on February 8, 2018(JavaScript required) .
- ↑ wacker.com: SILRES® BS 16 - Wacker Chemie AG , accessed on June 10, 2017.
