Monomer
Monomers ( ancient Greek μόνος monos 'one', 'single' and μέρος meros 'part', 'part') are low-molecular, reactive molecules that can combine to form unbranched or branched polymers . Monomers can be individual substances, but also mixtures of different compounds. In the first case homopolymers are formed , in the second copolymers ( heteropolymers ).
chemistry
| Typical monomers for plastics |
|---|
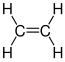 Ethene (ethylene) |
 Propene (propylene) |
 Vinyl chloride (VC) |
 Acrylamide |
In polymer chemistry , monomers are in principle all chemical compounds in which polymers can be formed through polyreactions . Monomers are often molecules with a C = C double bond or with at least two functional groups per molecule. Ring-shaped structures such as caprolactam can also be used as monomers for polyreactions. Monomers can be linked to polymers by chain polymerization , polycondensation or polyaddition .
There are also inorganic monomers, e.g. B. the orthosilicic acid H 4 SiO 4 , which polycondense to polysilicic acids, see silicas .
biochemistry
In biochemistry , the word "monomer" occasionally describes a subunit (also called a subunit ) as an individual component of a complex that is composed of several components. In connection with protein complexes , protein subunits are meant that are non-covalently linked to one another. Under suitable conditions, the subunits can oligomerize or associate or aggregate via intermediate stages ( dimer , trimer , tetramer , ...) .
See also
- Suffix -mer
- Constitutional repeat unit
Individual evidence
- ↑ Entry on monomeric . In: IUPAC Compendium of Chemical Terminology (the “Gold Book”) . doi : 10.1351 / goldbook.M04017 Version: 2.3.1.
- ↑ Entry on monomer molecule . In: IUPAC Compendium of Chemical Terminology (the “Gold Book”) . doi : 10.1351 / goldbook.M04019 Version: 2.3.1.
- ↑ Entry on monomers. In: Römpp Online . Georg Thieme Verlag, accessed on June 13, 2014.
- ^ MD Lechner, K. Gehrke and EH Nordmeier: Makromolekulare Chemie , 4th edition, Birkhäuser Verlag, 2010, pp. 48–170, ISBN 978-3-7643-8890-4 .
- ↑ a b Keyword sub-unit in: Hans-Dieter Jakubke, Ruth Karcher (Ed.): Lexikon der Chemie , Spektrum Akademischer Verlag, Heidelberg, 2001.