Senecioic acid
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
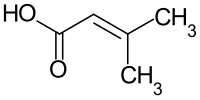
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Senecioic acid | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 5 H 8 O 2 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 100.12 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
0.96 g cm −3 (at 75 ° C) |
|||||||||||||||
| Melting point |
65-70 ° C |
|||||||||||||||
| boiling point |
194-195 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Senecio acid ( 3-methylcrotonic acid ) is an unsaturated and short-chain monocarboxylic acid from the group of alkenoic acids .
It is made from mesityl oxide by reaction with potassium hypochlorite , the methyl group adjacent to the keto group being oxidized . The carboxylic acid is released from the resulting potassium salt by adding sulfuric acid .
Individual evidence
- ↑ a b c Entry on 3-methylcrotonic acid in the GESTIS substance database of the IFA , accessed on November 7, 2019 (JavaScript required)
- ↑ a b Data sheet 3-Methylcrotonic acid from Sigma-Aldrich , accessed on July 28, 2019 ( PDF ).
- ↑ Lee Irvin Smith, WW Prichard, Leo J. Spillane: β, β-Dimethylacrylic acid In: Organic Syntheses . 23, 1943, p. 27, doi : 10.15227 / orgsyn.023.0027 ; Coll. Vol. 3, 1955, p. 302 ( PDF ).
