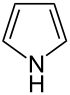
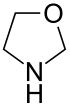
-in
The ending -idin is part of the name of numerous individual compounds and groups of substances in chemistry . In most cases, these are aromatic compounds that contain nitrogen . However, the naming does not follow a consistent system.
Derivatives of aromatic parent compounds
Numerous aromatic compounds derive their name from a parent compound. An amino group is introduced here as an additional substituent . Usually the ending -ol is separated and the ending -idin is added.
This is how:
- from toluene the Tolu idin
- of anisole (methoxybenzene), the anise idin ,
- from phenetole (ethoxybenzene) the Phenet idin
- from xylene the Xyl idin
- from cumene (isopropylbenzene) the Cum idin
- from mesitylene the Mes idin
- from pseudocumene the pseudocum idin
- of cresol the Kres idin
Phenol and benzene do not follow this system.
- Of phenol can be according to their names Phen idin form. This name would then have to be used for the aminophenol . However, the name phenidine is used as another common name for phenacetin .
- The name benzidine can accordingly be formed from benzene . This name would then have to be used for the aminobenzene (= aniline ). However, the name benzidine is used for 4,4'-diaminobiphenyl .
Nitrogen-containing heterocyclic compounds
Numerous nitrogen-containing heterocyclic compounds also contain the ending -idin such. B. pyridine , pyrimidine , lutidine , collidine , etc. Piperidine is a saturated heterocycle.
According to the Hantzsch-Widman system , this ending is part of the ending of nitrogen-containing, saturated rings with a ring size of three, four and five atoms. The endings -iridine (for three rings), -etidine (for four rings) and -olidine (for five rings) are used for these.
Nucleosides
The trivial ending -idin indicates that the nucleosides in question contain pyrimidine bases as a structural element, e.g. B .:
Compounds without nitrogen
There are, however, substances that end in -idin that do not contain nitrogen, e.g. B. hesperidin .
Individual evidence
- ↑ Philipp Fresenius and Klaus Görlitzer: Organic-chemical nomenclature , 3rd edition, Wissenschaftliche Verlagsgesellschaft, Stuttgart 1991, ISBN 3-8047-1167-7 , p. 295.