Pyrimidine
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
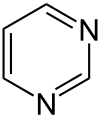
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Pyrimidine | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 4 H 4 N 2 | |||||||||||||||
| Brief description |
colorless to orange compound |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 80.09 g mol −1 | |||||||||||||||
| Physical state |
solid to liquid |
|||||||||||||||
| density |
1.016 g cm −3 (25 ° C) |
|||||||||||||||
| Melting point |
20-22 ° C |
|||||||||||||||
| boiling point |
123-124 ° C |
|||||||||||||||
| Vapor pressure |
19 hPa (22 ° C) |
|||||||||||||||
| solubility |
miscible with water, alcohol and ether |
|||||||||||||||
| Refractive index |
1.504 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Thermodynamic properties | ||||||||||||||||
| ΔH f 0 |
145.9 kJ mol −1 (liquid) |
|||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Pyrimidine is a six-membered heterocyclic aromatic amine with two nitrogen atoms , which is also known as 1,3-diazine according to the systematic nomenclature . The name is derived from the Greek pyr = heat, fire (because of the relationship with pyridine ) and the amidines . Pyrimidine is the base of the pyrimidine group .
structure
As an aza analogue (azalog) of pyridine, pyrimidine is one of the heteroaromatic diazines .
Manufacturing
Pyrimidine was first synthesized from barbituric acid by Siegmund Gabriel and James Colman at the end of the 19th century . This was reacted with phosphorus oxychloride to give 2,4,6-trichloropyrimidine. Dehalogenation with zinc powder resulted in the target compound. Dehalogenations were also carried out with 2,4-dichloropyrimidine and tetrachloropyrimidine.
More than 50 years after Gabriel's discovery, Hellmut Bredereck's working group developed a synthesis from C 3 building blocks and formamide . The tetraacetal, 1-methoxy-1,3,3-triethoxypropane, the enol ether 1,3,3-triethoxypropene and the enamine 3-diethylaminopropenal were used as a substitute for the unstable malondialdehyde (propanedial).
properties
Pyrimidine forms colorless crystals with a characteristic odor, which melt at temperatures above 20–22 ° C. Above the melting point there is a colorless liquid with a normal pressure boiling point of 124 ° C. The heat of vaporization is 49.89 kJ mol −1 . The enthalpy of combustion was determined to be −2288.9 kJ mol −1 , the enthalpy of formation to be 143.2 kJ mol −1 . The compound is easily soluble in water. It forms salts with acids.
literature
- Brockhaus ABC Chemie, FA Brockhaus Verlag Leipzig 1971.
- JA Joule, GF Smith, Heterocyclic Chemistry, Second Edition, Van Nostrand Reinhold Company, London, 1978, ISBN 0-442-30212-6 .
Individual evidence
- ↑ a b c d e f Entry on pyrimidine in the GESTIS substance database of the IFA , accessed on January 9, 2019(JavaScript required) .
- ↑ a b c d Entry on pyrimidine. In: Römpp Online . Georg Thieme Verlag, accessed on December 9, 2014.
- ^ The Merck Index . An Encyclopaedia of Chemicals, Drugs and Biologicals . 14th edition, 2006, ISBN 978-0-911910-00-1 , p. 1374.
- ↑ Data sheet Pyrimidines from Sigma-Aldrich , accessed on April 22, 2011 ( PDF ).
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Standard Thermodynamic Properties of Chemical Substances, pp. 5-25.
- ^ S. Gabriel, James Colman, About the Pyrimidine, Reports of the German Chemical Society, 32: 1525-1538 (1899); doi: 10.1002 / cber.18990320231 .
- ^ The Merck Index, 11th Edition, Merck and Co., Rahway, USA, 1989, p. 1270.
- ↑ H. Bredereck, R. Gompper, G. Morlock: Formamide reactions, VIII. A new pyrimidine synthesis , Chemical Reports 90 , 942-952 (1957); doi: 10.1002 / cber.19570900613 .
- ↑ H. Bredereck, R. Gompper, H. Herlinger: Formamid reactions, XI. Presentation, properties and reactions of pyrimidine , Chemischeberichte, 91 , 2832-2849 (1958); doi: 10.1002 / cber.19580911240 .
- ↑ CRC Handbook of Data on Organic Compounds, 2nd Edition, Weast, RC and Grasselli, JG, ed (s)., CRC Press, Inc., Boca Raton, FL, 1989, 1.
- ↑ a b Nabavian, PM; Sabbah, R .; Chastel, R .; Laffitte, M., Thermodynamique de composes azotes. II. Etude thermochimique des acidic aminobenzoiques, de la pyrimidine, de l'uracile et de la thymine., J. Chim. Phys., 1977, 74, pp. 115-126.




