1,2-diiodopropane
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
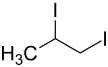
|
|||||||||||||
| Mixture of stereoisomers - structural formula without stereochemistry | |||||||||||||
| General | |||||||||||||
| Surname | 1,2-diiodopropane | ||||||||||||
| Molecular formula | C 3 H 6 I 2 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 295.8 g mol −1 | ||||||||||||
| Physical state |
liquid |
||||||||||||
| density |
2.490 g cm −3 |
||||||||||||
| Melting point |
−20 ° C |
||||||||||||
| boiling point |
227 ° C |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
1,2-Diiodopropane is a chemical compound that belongs to the halogen alkanes . It is isomeric to 1,1-diiodopropane , 1,3-diiodopropane and 2,2-diiodopropane .
presentation
1,2-Diiodopropane can be produced by the electrophilic addition of iodine to propene . Since the compound is relatively unstable, the reaction can easily be reversed.
properties
The critical temperature of 1,2-diiodopropane is 780.49 K , the critical pressure 42.06 bar . The enthalpy of vaporization at the boiling point is 42.844 kJ / mol.
Individual evidence
- ↑ Dictionary of organic compounds, p. 2500 ( limited preview in Google book search).
- ↑ a b c Carl L. Yaws: "Thermophysical properties of chemicals and hydrocarbons", p. 8 ( limited preview in the Google book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ Thomas M. Schmitt: "Analysis of surfactants", p. 67 ( limited preview in the Google book search).
- ^ Carl L. Yaws: "Thermophysical properties of chemicals and hydrocarbons", p. 315 ( limited preview in the Google book search).
