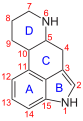
1P-ETH-LAD
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| General | ||||||||||
| Surname | 1P-ETH-LAD | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 24 H 31 N 3 O 2 | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| Drug information | ||||||||||
| Mechanism of action | ||||||||||
| properties | ||||||||||
| Molar mass | 393.53 g mol −1 | |||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
1P-ETH-LAD (English 1- propionyl- eth yl - nor- l ysergic a cid d iethylamide ) is a chemically produced derivative of lysergic acid , which occurs naturally as an ergot alkaloid , and an analogue of LSD , ETH-LAD and 1P -LSD . Even in very small doses , it produces long-lasting hallucinogenic effects. Pharmacologically, 1P-ETH-LAD belongs to the group of serotonin-related psychedelic substances.
chemistry
Chemically, 1P-ETH-LAD belongs to the Ergoline structural class . The tetracyclic ergoline is characteristic of the chemical structure of ergot alkaloids . The ergolines found in nature are methylated on the nitrogen in position 6 of the ergoline system . In contrast to ETH-LAD, 1P-ETH-LAD has an additional N 1 - propionyl group and, in contrast to 1P-LSD, an N 6 - ethyl group instead of an N 6 - methyl group .
pharmacology
1P-ETH-LAD hydrolyses (in the same way or similar to 1P-LSD ) in the body, splitting off propionic acid to form ETH-LAD, as an LC-MS analysis showed that ETH-LAD is formed when 1P-ETH-LAD is in human blood serum is incubated. It is therefore assumed that the profile will be similar to that of ETH-LAD.
literature
- Simon D. Brandt, Pierce V. Kavanagh et al. a .: Return of the lysergamides. Part III: Analytical characterization of N 6 -ethyl-6-norlysergic acid diethylamide (ETH-LAD) and 1-propionyl ETH-LAD (1P-ETH-LAD). In: Drug Testing and Analysis. 2017, doi : 10.1002 / dta.2196 .
- Lea Wagmann, Lilian HJ Richter a. a .: In vitro metabolic fate of nine LSD-based new psychoactive substances and their analytical detectability in different urinary screening procedures. In: Analytical and Bioanalytical Chemistry. 2019, doi : 10.1007 / s00216-018-1558-9 .
See also
- METH-LAD (LSD)
- 1P-METH-LAD (1P-LSD)
- PRO-LAD
Web links
- isomerdesign.com: 1P-ETH-LAD (English)
Individual evidence
- ↑ a b Simon D. Brandt, Pierce V. Kavanagh, Folker Westphal, Alexander Stratford, Simon P. Elliott, Khoa Hoang, Jason Wallach, Adam L. Halberstadt: Return of the lysergamides. Part I: Analytical and behavioral characterization of 1-propionyl-d-lysergic acid diethylamide (1P-LSD). In: Drug Testing and Analysis. 2015, pp. 891–902, doi : 10.1002 / dta.1884 .
- ^ A b F. Schifano, L. Orsolini, D. Papanti, J. Corkery: NPS: Medical Consequences Associated with Their Intake. In: Current Topics in Behavioral Neurosciences. Volume 32, 2017, pp. 351-380, doi : 10.1007 / 7854_2016_15 , PMID 27272067 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Lea Wagmann, Lilian HJ Richter a. a .: In vitro metabolic fate of nine LSD-based new psychoactive substances and their analytical detectability in different urinary screening procedures. In: Analytical and Bioanalytical Chemistry. , doi : 10.1007 / s00216-018-1558-9 .
- ↑ Simon D. Brandt, Pierce V. Kavanagh et al. a .: Return of the lysergamides. Part III: Analytical characterization of N 6 -ethyl-6-norlysergic acid diethylamide (ETH-LAD) and 1-propionyl ETH-LAD (1P-ETH-LAD). In: Drug Testing and Analysis. 2017, doi : 10.1002 / dta.2196 .