2,3-dibromohydroquinone
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| General | ||||||||||
| Surname | 2,3-dibromohydroquinone | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 6 H 4 Br 2 O 2 | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 267,90 g · mol -1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
184 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
2,3-Dibromohydroquinone is a chemical compound that belongs to both the polyphenols and the halogen aromatic compounds . Together with 2,5-dibromohydroquinone and 2,6-dibromohydroquinone, it is one of the three positionally isomeric dibromohydroquinones.
presentation
2,3-Dibromohydroquinone can be made by bromination of 1,4-benzoquinone with elemental bromine .
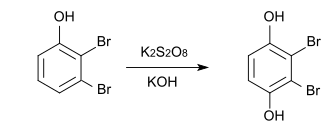
Synthesis by Elbs oxidation from 2,3-dibromophenol is also possible.
Reactions
Esterification with acetic anhydride results in the diacetate, which is registered under CAS number 40101-04-0 and has a melting point of 163 ° C.
Oxidation with iron (III) chloride leads to 2,3-dibromo-1,4-benzoquinone (melting point 124–125 ° C).
Individual evidence
- ^ A b J. Buckingham: Dictionary of organic compounds, Volume 9, p. 1914 ( limited preview in the Google book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ E. Sarauw: Benzenequinone and derivatives of the same , quarterly journal of the Natural Research Society in Zurich, 1881.
- ↑ JF Bagli, P. L'Ecuyer: Arylation of Quinones by Diazonium Salts - VII. Synthesis and Structure of some Aryl-Chloro-p-Benzoquinones . In: Canadian Journal of Chemistry . 39 (5), 1961, pp. 1037-1048, doi : 10.1139 / v61-128 .